Plastic manufacturing consumes a lot of energy and emits greenhouse gases like carbon dioxide, which are harmful to the environment and atmosphere and contribute significantly to global warming. The majority of plastic packaging that is sent to landfills around the world ends up in our natural environments as a result of poor waste management systems. Every year, 8 million tons of plastic end up in the ocean. Plastic pollution endangers wildlife, disrupts ecosystems, and endangers human health.
A research team is working on polymers that can be broken down into their constituent parts; as a result, when the catalyst for depolymerization is absent or removed, the polymers will be highly stable, and their thermal and mechanical properties can be tuned to meet the needs of various applications.
Plastics sustainability has advanced significantly in recent years, thanks in large part to scientific advances. Despite the fact that plastics are becoming more environmentally friendly, the world continues to be polluted because many industries rely on them for widely used products.
Plastic pollution has become one of the most pressing environmental issues, as the rapidly increasing production of disposable plastic products overwhelms the world’s ability to deal with them.
Dr. Junpeng Wang, assistant professor in the UA’s School of Polymer Science and Polymer Engineering, has developed a solution to reduce such waste and pave the way for a more sustainable future that can appeal to the rubber, tire, automobile, and electronics industries. Despite the fact that this work is supported by the University of Arizona, Wang recently received a National Science Foundation CAREER Award, which will help fund future developments from this research.
The issue: Synthetic polymers, such as rubber and plastics, are used in almost every aspect of daily life. The dominance of synthetic polymers is largely due to their excellent mechanical properties and versatility. However, because of their high durability, waste materials containing these polymers have accumulated in the land and oceans, posing serious environmental concerns.
Furthermore, because over 90% of these polymers are derived from finite natural resources such as petroleum and coal, their production is unsustainable if they cannot be recycled and reused.
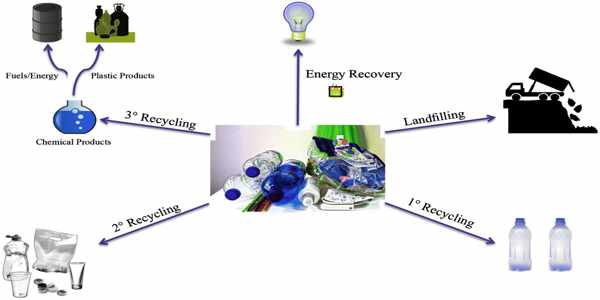
To achieve a circular use of materials, one promising solution to address the challenges of plastics sustainability is to replace current polymers with recyclable ones. Despite recent advances, few recyclable polymers have excellent thermal stability and high-performance mechanical properties of traditional polymers. Wang and his team’s recyclable materials are unique in their superior thermal stability and versatile mechanical properties. Their paper, “Olefin Metathesis-Based Chemically Recyclable Polymers Enabled by Fused-Ring Monomers,” was published in Nature Chemistry last week.
“We’re particularly interested in chemically recyclable polymers that can be broken down into their constituents (monomers),” Wang says. “The recycled monomers can be reused to produce the polymers, allowing for a circular use of materials that not only helps to preserve the finite natural resources used in the production of plastics but also addresses the issue of unwanted end-of-life accumulation of plastic objects.”
The identification of the appropriate monomer is critical in the design of chemically recyclable polymers. The researchers discovered a targeting monomer through careful computational calculations. They then used readily available starting materials to create the monomer and polymers via chemical synthesis.
Wang’s research group, which includes graduate students in polymer science and a postdoctoral scientist, aims to address these challenges by developing polymers that can be broken down into their constituent parts. When the depolymerization catalyst is absent or removed, the polymers are highly stable, and their thermal and mechanical properties can be tuned to meet the requirements of various applications.
“The chemically recyclable polymers we developed have excellent thermal stability and robust mechanical properties, and they can be used to prepare both rubber and plastics,” Wang explains. “We anticipate that this material will be a viable replacement for current polymers. Our molecular design is guided by computation, demonstrating the transformative power of combining computational and experimental work. When compared to other recyclable polymers that have been demonstrated, the new polymers we demonstrate have much higher mechanical stability and versatility. With the addition of a catalyst, the polymer can be degraded into its constituent monomer for recycling.”
Wang’s research group’s next goal is to broaden the scope of chemically recyclable polymers and develop carbon-fiber-reinforced polymer composites. The team will also conduct an economic analysis of this industrial process as well as a life-cycle analysis for the commercialization of the polymers.
















