According to new findings, researchers created a bionic arm for patients with upper-limb amputations that allows wearers to think, behave, and function like a person without an amputation. The bionic system integrates three critical functions: intuitive motor control, touch and grip kinesthesia, and the intuitive feeling of opening and closing the hand.
According to new findings published in Science Robotics, Cleveland Clinic researchers created a first-of-its-kind bionic arm for patients with upper-limb amputations that allows wearers to think, behave, and function like a person without an amputation.
The bionic system was created by an international research team led by the Cleveland Clinic that combines three important functions: intuitive motor control, touch and grip kinesthesia, and the intuitive feeling of opening and closing the hand. The University of Alberta and the University of New Brunswick were among the collaborators.
“We modified a standard-of-care prosthetic with this complex bionic system, which enables wearers to move their prosthetic arm more intuitively and feel sensations of touch and movement at the same time,” said lead investigator Paul Marasco, Ph.D., associate professor in the Department of Biomedical Engineering at Cleveland Clinic Lerner Research Institute. “These findings are an important step toward restoring complete natural arm function to people who have had amputations.”
A bionic arm for patients with upper-limb amputations that allows wearers to think, behave and function as a person without an amputation, according to new findings.
The system is the first to test all three sensory and motor functions of a neural-machine interface in a prosthetic arm simultaneously. The neural-machine interface is linked to the wearer’s limb nerves. It allows patients to send nerve impulses from their brains to the prosthetic when they want to use or move it, as well as to receive physical information from their surroundings and relay it back to their brain via their nerves.
The bi-directional feedback and control of the artificial arm allowed study participants to perform tasks with the same level of accuracy as non-disabled people.
“Perhaps what we were most excited to learn was that they made judgments, decisions, calculated and corrected for their mistakes like a person without an amputation,” said Dr. Marasco, the Laboratory for Bionic Integration’s director. “People acted as if they had a natural hand with the new bionic limb. Normally, these brain behaviors differ greatly between people who have and do not have upper limb prosthetics.”
Dr. Marasco also has an appointment to in Cleveland Clinic’s Charles Shor Epilepsy Center and the Cleveland VA Medical Center’s Advanced Platform Technology Center.
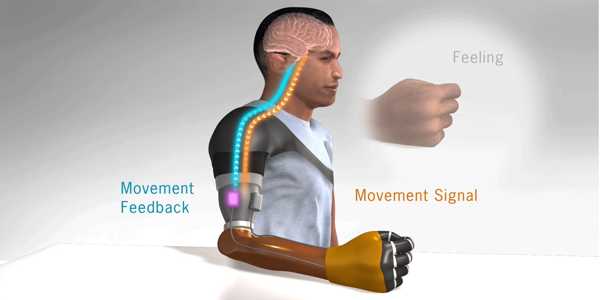
The researchers tested their new bionic limb on two study participants who had previously undergone targeted sensory and motor reinnervation – procedures that create a neural-machine interface by redirecting amputated nerves to remaining skin and muscles. Due to the small size of the study, additional research will be required.
Touching the skin with small robots activates sensory receptors, allowing patients to perceive the sensation of touch in targeted sensory reinnervation. When patients think about moving their limbs during targeted motor reinnervation, the reinnervated muscles communicate with a computerized prosthesis to move in the same way. Furthermore, small, powerful robots vibrate kinesthetic sensory receptors in those same muscles, providing prosthesis wearers with the sensation that their hand and arm are moving.
Participants wore the advanced prosthetic and performed tasks resembling basic, everyday behaviors that necessitate hand and arm functionality. The researchers used newly developed advanced evaluation tools to compare the performance of the bionic limb to that of non-disabled people and people with amputations who use traditional prosthetic devices. They also compared how people with advanced prosthetics fared when all three sensory and motor modalities were enabled at the same time versus separately.
According to Dr. Marasco, because people who wear traditional prosthetics cannot feel their limbs, they behave differently than people who do not have an amputation when performing daily tasks. Traditional prosthesis wearers, for example, must constantly monitor their prosthetic while using it and struggle to learn to correct errors when they apply too much or too little force with their hand.
The researchers were able to see, using the new artificial arm and advanced evaluation tools, that the study participants’ brain and behavioral strategies changed to match those of a person who had not had an amputation. They no longer had to keep an eye on their prosthesis, they could find things without looking, and they could correct their mistakes more effectively.
“Prosthetics advancements over the last decade or two have helped wearers achieve better functionality and manage daily living on their own,” Dr. Marasco said. “For the first time, people with upper limb amputations can ‘think’ like an able-bodied person, which has the potential to offer prosthesis wearers new levels of seamless reintegration back into daily life.”
Beyond this study, the international team’s new outcome measurements for brain behavior and functionality developed to evaluate the bionic system can be applied to any upper limb prosthetic or deficit that involves sensation and movement.