Improvements in water-splitting reactions frequently focus on increasing efficiency, lowering energy consumption, and using environmentally friendly materials. Due to the low electrical conductance of (oxy)hydroxide species produced in situ, electrochemical catalysts used in water splitting frequently perform poorly. To address this issue, scientists have created an electrode with a Schottky junction formed at the interface of metallic Ni-W5N4 and semiconducting NiFeOOH. The proposed electrode has excellent catalytic activity and can support continuous industrial seawater splitting for 10 days.
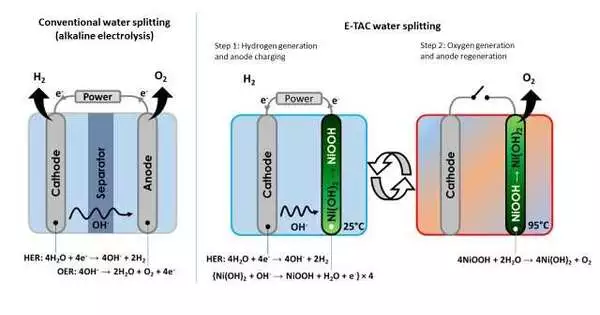
Green hydrogen (or H2) derived from renewable energy sources is the fuel of the future. One of the most common methods of producing green H2 is electrolysis, or the splitting of water into oxygen and hydrogen using an electrochemical cell. It is a simple reaction that produces high-quality products while emitting no carbon dioxide.
Despite its benefits, electrochemical water splitting has yet to gain traction on a commercial scale. This is because active (oxy)hydroxide catalysts generated in situ during electrochemical processes have low electrical conductivity. This, in turn, limits catalytic activity, impeding both hydrogen and oxygen evolution reactions in the cell.
Our research utilized this potential energy barrier present in the Schottky junction to accelerate electron flow in the electrode, leading to a significant increase in oxygen evolution reaction activity, and expediting overall water splitting.
Dr. Seo
The issue of (oxy)hydroxide’s poor electrical properties has long been a barrier to achieving efficient water splitting. Now, a team of researchers led by Associate Professor Junhyeok Seo of Gwangju Institute of Science and Technology’s Department of Chemistry has discovered a solution to this problem in the form of Schottky junctions.
They demonstrated an electrode with Schottky junction formed at the interface of metallic nickel-tungsten nitride (Ni-W5N4) and semiconducting n-type nickel-iron (oxy)hydroxide (NiFeOOH) catalyst in a recent study made available online on 30 August 2023 and to be published in Volume 340 of the Applied Catalysis B: Environmental journal in January 2024. This electrode was able to overcome the (oxy)hydroxide conductance limit and improve the setup’s water-splitting ability.
Notably, two materials, a metal, and a semiconductor, with largely different electronic behaviors were put in contact to make an energy difference at the interface, forming a junction. “Our research utilized this potential energy barrier present in the Schottky junction to accelerate electron flow in the electrode, leading to a significant increase in oxygen evolution reaction activity, expediting overall water splitting,” explains Dr. Seo, highlighting the core mechanism behind their newly designed electrode.
Using electrocatalytic water splitting, the researchers discovered that the Ni-W5N4 alloy catalyzed the hydrogen evolution reaction, resulting in a current density of 10 mA/cm2 at a small overpotential of 11 mV. Furthermore, the rectifying Schottky junction formed at the Ni-W5N4|NiFeOOH interface nullified the non-conductive lamination produced by (oxy)hydroxide species. It had a forward bias current density of 11 mA/cm2 at 181 mV overpotential. The electrode’s electrochemical analysis revealed that the improved catalytic activity was indeed due to the Schottky junction.
Finally, the researchers created an electrolyzer for industrial seawater electrolysis using their Schottky junction electrode. They discovered that the new device could run continuously for 10 days while also displaying exceptional catalytic activity and durability during electrolysis. It showed a remarkable current density of 100 mA/cm2 at an overpotential of just 230 mV.
Overall, the researchers believe that their findings can help to develop a sustainable hydrogen production strategy that will eventually replace traditional methods that still rely on fossil fuels. “Freshwater and seawater are abundant and renewable sources of protons,” Dr. Seo concludes. Efficient water splitting systems ensure that we can establish long-term production of zero-carbon hydrogen fuel, thereby assisting in the management of our current climate problems.”
Let us hope that the successful demonstration of a Ni-W5N4|NiFeOOH electrode for water splitting opens up new opportunities for Schottky junction-based high-performance energy storage and conversion systems!