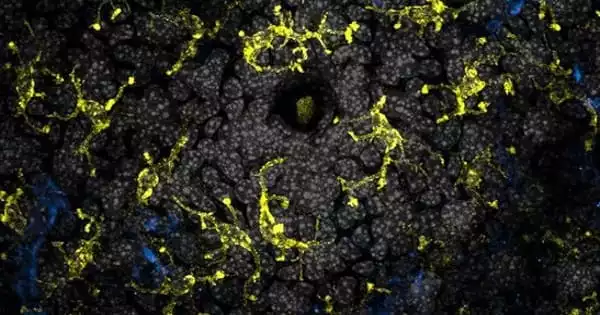
Animals who move to the city may get more than they bargained for. Gut microbes from humans in cities may be contaminating urban wildlife, putting the animals’ health at danger. The concept of One Health emphasizes the ecological links between human, animal, and environmental health. To date, much of the One Health literature has focused on the transmission of infections from animals (e.g., developing zoonoses) and the environment to humans.
Fecal samples from humans and animals around the world reveal that urban critters have microbial communities that are far more similar to those found in urban humans than in rural people and wildlife, according to researchers in a preliminary study published on bioRxiv.org. While previous research has discovered that captive animals can acquire human microbes, some of which have been linked to gastrointestinal disorders, immune deficiencies, and even stunted growth, this is the first time a humanizing effect on wildlife has been discovered in cities, and between humans and reptiles.
Microbiota boost the immune system, break down potentially hazardous dietary molecules, and manufacture vitamins and amino acids such as B vitamins and vitamin K. The main enzymes required to produce vitamin B12, for example, are exclusively present in bacteria and not in plants or mammals.
Many of the microorganisms found in industrialized human populations are also found in urban wildlife populations, but they are virtually absent in rural populations, according to Andrew Moeller, an evolutionary biologist at Cornell University.
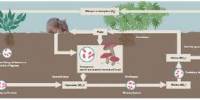
The most basic explanation would be that the animals are just cohabiting in the same place and hence picking up microorganisms in their surroundings. It could also be the result of an urban gastronomical lifestyle, as some bacteria are related with high-fat, high-protein meals commonly consumed in cities.
Taichi Suzuki
Many animals have symbiotic microbe communities, which are evolutionary products that play a significant role in immunity and fitness. Moeller and his colleagues examined 492 fecal samples collected from humans, coyotes, and lizards in urban and rural settings as diverse as Edmonton, Canada, and Amazonian towns in Venezuela to discover how microbial communities in urban animals compared to those in humans. The researchers employed genomic analysis to determine the amount of microbial DNA in each sample before comparing the microbial profiles of urban and rural hosts.
The researchers discovered that urban lizards and coyotes had gut microbiome populations that were more similar to urban humans than to rural humans or wildlife. They detected 18 bacterium lineages in urban wildlife that did not exist in their rural counterparts.
According to Taichi Suzuki, an evolutionary biologist at the Max Planck Institute for Developmental Biology in Tübingen, Germany, who was not involved in the study, the parallel changes in microorganisms seen in diverse animals from different locations are convincing patterns of urban-specific microbial changes. “A key question is the mechanism generating this trend, which needs to be looked into more,” he says.
According to Moeller, the human gut is a source of the new bacteria lineages identified in urban wildlife, thus they probably took them up from people. As we go about our regular lives, we shed germs from our personal microbial clouds. “The most basic explanation would be that the animals are just cohabiting in the same place and hence picking up microorganisms in their surroundings,” adds Moeller. It could also be the result of an urban gastronomical lifestyle, as some bacteria are related with high-fat, high-protein meals commonly consumed in cities.
Diet, in addition to family genes, environment, and medication use, influences the types of microbiota that live in the colon. All of these elements contribute to the creation of a distinct microbiome from person to person. A high-fiber diet, in particular, has an impact on the kind and quantity of microbiota in the intestines. Dietary fiber can only be broken down and fermented by microbial enzymes in the colon. Fermentation results in the release of short chain fatty acids (SCFA).
This lowers the pH of the colon, determining the sort of microbiota that may thrive in this acidic environment. Some dangerous bacteria, such as Clostridium difficile, are inhibited by the lower pH. SCFA research is expanding to investigate their wide-ranging effects on health, such as increasing immune cell activity and maintaining appropriate blood glucose and cholesterol levels.
According to Mark van der Giezen, a microbiologist at the University of Stavanger in Norway who was not part in this study, the research is a good primer but might benefit from taking into account local cuisines and illnesses. “Urbanization is a highly complex sociological phenomena,” he explains.
The microbiome is a living, dynamic ecology in which the relative abundance of species can change on a daily, weekly, and monthly basis depending on nutrition, medicine, activity, and a variety of other environmental exposures. However, scientists are still in the early stages of comprehending the microbiome’s extensive involvement in health and the scope of issues that can arise when normal interactions between the microbiome and its host are disrupted.