A new study led by Harvard Medical School researchers has discovered a set of cellular receptors for at least three related alphaviruses that are shared by mosquitos, humans, and animals that host the virus.
Going a step further, the researchers tested a “decoy” molecule in a series of experiments in cells and animal models, successfully preventing infection and slowing disease progression, an important first step toward developing preventive and curative medicines against these highly pathogenic viruses with pandemic potential. The findings were published in the journal Nature.
Understanding the basic biology of a virus’s life cycle is critical to finding a way to prevent an illness, and developing such foundational knowledge prior to an outbreak is critical for preparing for future outbreaks, according to study senior author Jonathan Abraham, assistant professor of microbiology at HMS and an infectious disease specialist at Brigham and Women’s Hospital.
“Knowing how a virus enters and infects a cell is as fundamental as it gets,” he says. “Viral entry into human or other mammalian cells marks the beginning of infection and, eventually, disease, and is an excellent place to look for potential preventive and curative strategies.”
The alphaviruses studied by the researchers, including EEEV, have a history of causing lethal, albeit brief, outbreaks, but little is known about how the virus attacks host cells. Only a few other receptors associated with alphavirus infection have been identified. According to Abraham, one of the reasons for the lack of targeted treatments for these lethal viruses is a knowledge gap.
Knowing how a virus enters and infects a cell is as fundamental as it gets. Viral entry into human or other mammalian cells marks the beginning of infection and, eventually, disease, and is an excellent place to look for potential preventive and curative strategies.
Jonathan Abraham
Infected mosquitoes
Eastern equine encephalitis (triple E) is typically transmitted to humans through the bite of an infected mosquito. The most recent triple E outbreak occurred in New England in 2019. According to a 2019 triple E outbreak report from the Centers for Disease Control and Prevention, the triple E outbreak hospitalized all 32 known cases and killed 12 of those infected.
The virus has a 30% fatality rate, which means it kills nearly one-third of those infected, comparable to Ebola virus disease or smallpox. According to the CDC, nearly half of those who survive have long-term neurologic complications from the disease. Larger outbreaks occurred in the 1930s and 1950s, but data collection and diagnostics have changed so much since then that comparing the magnitude of the outbreaks is difficult, according to the CDC report.
There are significant advantages to conducting this research prior to the onset of major outbreaks, according to Abraham, who has worked on identifying receptors that viruses use as pathways for entering cells and causing disease, as well as developing antibody treatments to prevent SARS-CoV-2 infection and COVID-19 infection.
Prior work on SARS-CoV during the early 2000s SARS outbreak, for example, was critical to increasing preparedness against SARS-CoV-2. Because the closely related viruses that cause COVID-19 and SARS both attack human hosts by using the same receptor on human cells to gain entry and cause disease, the availability of SARS-CoV-2 genome sequences within days of the novel virus’s initial reporting was critical to the rapid development of vaccines and antibody treatments for COVID-19.
According to Abraham, new screening tools and techniques in molecular biology, protein biochemistry, biophysics, and structural biology provide unprecedented power to learn more about the basic biology of viruses before they emerge as global threats. “The time to prepare for these uncertain but potentially catastrophic scenarios is well before they occur,” Abraham said.
Gene-editing screen
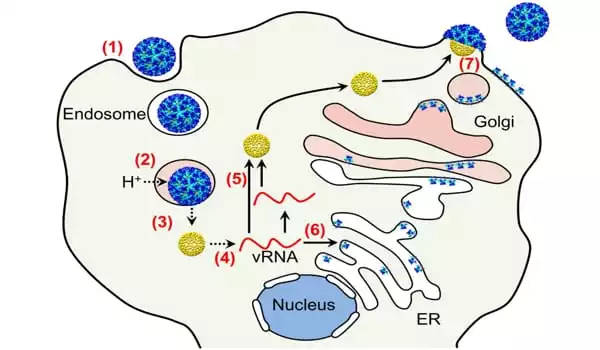
The researchers first used a CRISPR-Cas9 gene-editing screen to identify a receptor for Semliki Forest virus (SFV) on human cells for the current study. In rodents and other animals, SFV is an alphavirus that can cause severe neurologic disease and death. The SFV receptors discovered by the researchers were also compatible with EEEV and Sindbis, a related virus that causes fever and severe joint pain in humans and neurological disease in animals and rodents.
“That is why it is critical to study these viruses as families,” Abraham explained. “You could end up studying a virus like SFV and discovering something really exciting about the biology of related viruses, which has the potential to unlock novel ways to treat new categories of viruses capable of causing serious disease and outbreaks in humans.”
Identifying a receptor for multiple viruses would allow scientists and physicians to get a head start on developing tools to prevent, control, and treat infections in the event of a virus outbreak, according to Abraham.
To confirm that the receptors in question were important in causing infection, the researchers used a decoy protein, a molecule with a structure that mimics the receptor and can trick the virus into binding to the drug rather than the host cell it is attempting to infect. The molecule effectively disables the virus and prevents it from entering the host cell, thereby preventing infection.
Experiments by the team showed that preventing the virus from interacting with the host cell receptor prevented infection of human and mouse neurons. They also discovered that the decoy molecule protected infected mice from developing rapidly fatal alphavirus encephalitis, which suggests that this pathway could be targeted by drugs or antibodies to treat alphavirus encephalitis in humans when infections do occur. The researchers emphasize that their animal studies were conducted with Semliki Forest virus rather than EEEV, so more research is needed to confirm that the same approach would work for other alphaviruses and in humans.
It usually takes years to turn a simple insight like this into a clinical tool. Researchers must ensure that it is safe and effective, as well as determine the best way to administer the decoy molecules. It is critical to build this base of knowledge before the next pandemic to buy as much time as possible to prepare for emerging viruses, according to Abraham.
“All of us scientists, policymakers, and citizens would benefit greatly from learning to think more ahead,” Abraham said. “The more we understand about the basic biology of various virus families, particularly how they infect and interact with their hosts, the better prepared we will be to face whatever comes next.”