Extracting CO2 from the atmosphere is one of the most effective ways to reverse climate change without relying on expensive technologies, complicated tax schemes, or denying billions of people access to the energy they require to live a decent life.
A-CuTi@Cu, a new electrocatalyst, converts carbon dioxide (CO2) into liquid fuels. According to a team of Chinese researchers who published their findings in the journal Angewandte Chemie, active copper centered on an amorphous copper/titanium alloy efficiently produces ethanol, acetone, and n-butanol.
The majority of our global energy needs are still met by burning fossil fuels, which contributes to the greenhouse effect by releasing CO2. To combat global warming, we must look for ways to use CO2 as a raw material for basic chemicals. A climate-neutral, artificial carbon cycle could be established through electrocatalytic CO2 conversion using renewable energy. Excess energy generated by photovoltaics and wind energy could be stored by electrocatalytically producing fuels from CO2. These could then be disposed of as needed. Conversion to liquid fuels would be advantageous due to their high energy density and ease of storage and transportation. However, the electrocatalytic formation of products containing two or more carbon atoms (C2+) is extremely difficult.
A-CuTi@Cu, a new electrocatalyst, converts carbon dioxide (CO2) into liquid fuels. Using this approach, we prepared several new catalysts and discovered that they are all highly efficient in converting CO2 to other hydrocarbons. We intend to carry on this research in collaboration with industry in order to advance this promising technology.
Fei Hu, Tingting Kong, and Yujie Xiong from Foshan University
A team led by Fei Hu, Tingting Kong, Jun Jiang, and Yujie Xiong from Foshan University (Foshan, Guangdong), the University of Science and Technology of China (Hefei, Anhui), and Xi’an Shiyou University (Xi’an, Shaanxi has developed a novel electrocatalyst that efficiently converts CO2 to liquid fuels with multiple carbon atoms (C2-4). Ethanol, acetone, and n-butanol are the main byproducts.
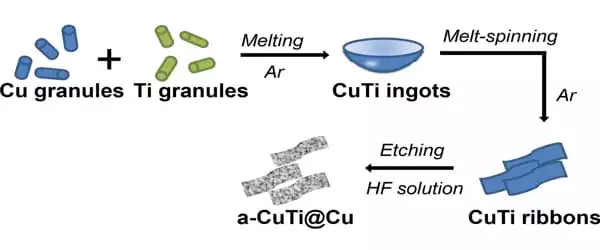
Thin ribbons of a copper/titanium alloy are etched with hydrofluoric acid to remove the titanium from the surface to create the electrocatalyst. This yields a material known as a-CuTi@Cu, which has a porous copper surface on an amorphous CuTi alloy. It has catalytically active copper centers with exceptional activity, selectivity, and stability for reducing CO2 to C2+ products (total faradaic efficiency of about 49 percent at 0.8 V vs. reversible hydrogen electrode for C2-4, and it is stable for at least three months). Pure copper foil, on the other hand, produces C1 products but almost no C2+ products.
A multistep electron-transfer process via various intermediates is involved in the reaction. The inactive titanium atoms beneath the surface play an important role in the new electrocatalyst, increasing the electron density of the Cu atoms on the surface. This stabilizes the adsorption of CO, a key intermediate in the formation of multicarbon products, allowing for a high coverage of the surface with CO and lowering the energy barrier for di- and trimerization of the *CO as new carbon-carbon bonds are formed.
“Using this approach, we prepared several new catalysts and discovered that they are all highly efficient in converting CO2 to other hydrocarbons,” Liu said. “We intend to carry on this research in collaboration with industry in order to advance this promising technology.”
Carbon Engineering’s Direct Air Capture system can currently remove a ton of CO2 from the atmosphere for around $100. Individual systems would be designed to capture approximately a million tons of CO2 per year, necessitating the installation of tens of thousands of systems to keep up with global emissions and reduce atmospheric CO2 to normal levels by 2040. There are just under 70,000 gas stations in the United States alone, which isn’t nearly enough to save the planet.
Carbon Engineering’s Air to Fuel technology then produces synthetic, liquid transportation fuels like gasoline, diesel, and Jet-A. To produce hydrocarbon fuels, the process combines CO2 captured from the atmosphere via their Direct Air Capture system with hydrogen.
A photoelectrochemical cell made of thin metal oxide films is used in this technology. The reaction is carried out using sunlight (primarily the ultraviolet (UV), visible, and infrared (IR) portions) and low-cost titanium dioxide composites. The device can be used to capture carbon dioxide from industrial processes before it is released into the atmosphere and convert it to a useful fuel like methane. These devices have low manufacturing and material costs, allowing them to be deployed in the commercial market. They can be made to be extremely compact and efficient, and they can be used in sensor and detector applications.