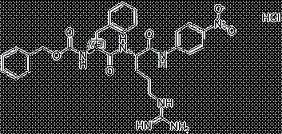
Abstract
The present study was undertaken to investigate that which product give better release profile from these two companies product. Metformin HCl is hydrophilic in nature. The USP paddle method was selected to perform the dissolution study. Release kinetics of metformin HCl from extended release tablets was studied in two dissolution media (0.1N HCl & phosphate buffer, pH=6.8) & examined that in which media, the drug is more released.
Statistically significant differences were found among the drug release profile from these two samples & the release data were treated in different mathematical models to identify the release mechanism. From these two samples, it was observed that drug release from metformin HCl XR tablets was occurred by floating control system.
It was observed that release of metformin HCl of sample-1 was less than the release of sample-2 in acidic media & also in phosphate buffer media. It was also observed that release of metformin HCl of sample-1 & sample-2 in basic media was more than in acidic media.
It was found that the release of metformin HCl followed the zero order kinetics & also showed the tendency to follow first order as well as Higuchi’s diffusion fashion. On the basis of results, it was showed in this study that the profile & kinetics of drug release of sample-2 was the best among market preparations.
2.0. Introduction:
Dissolution study was done with two samples of available market preparations & after that compared with another by calculating the percentages of release. Dissolution test was carried out in two medium such as 0.1N HCl for 2 hr & phosphate buffer (pH= 6.8) for 3 to 8 hrs. For this test, six vessel tablet dissolution tester PHARMA TEST Mo-del-DT 70 was used.
2.1. Dissolution Study for Metformin HCl 500 XR
Two samples are-
Sample no. 1
Brand name: Comet 500 XR
Company name: Square Pharmaceuticals ltd.
Sample no. 2
Brand name: Glucomet 500 XR
Company name: Aristopharma ltd.
2.2. Dissolution:
The time takes for the drug to dissolve from the dosage form is dissolution time. Numerous factors affect dissolution. Thus the dissolution medium, agitation, temperature are carefully controlled. The dissolution medium may be water, simulated gastric juice. The temperature is usually 37.5°C. The apparatus & specifications may be found in the U.S.P.
The U.S.P. methods are official however there is a wide variety of methods based on other apparatus. These are used because they may be faster, cheaper, easier, sensitive to a particular problem for a particular drug, or developed by a particular investigator.
Dissolution tests are used as quality control to measure variability between batches which may be reflected by in vivo performance. Thus the in vitro test may be a quick method of ensuring in vivo performance. Thus there has been considerable work aimed at defining the in vitro/in vivo correlation.
2.3. Physicochemical & physiological factors affecting drug dissolution in the gastrointestinal tract:
There are various physicochemical & physiological factors that affecting drug dissolution in gastrointestinal tract are given below:
Table: 2.1. Factors affecting drug dissolution.
| Factor | Physicochemical parameter | Physiological parameter |
| Effect of surface area of drug | Particle size, wet ability | Surfactants in gastric juice & bile |
| Solubility in diffusion layer | Hydrophilicity, crystal structure, solubilization | PH, buffer capacity, bile, food components |
| Amount of drug already dissolved | Permeability, transit | |
| Diffusivity of drug | Molecular size | Viscosity of luminal contents |
| Boundary later thickness | Motility patterns & flow rate | |
| Volume of solvent available | Gastrointestinal secretions, co-administered fluids |
2.4. Drug Dissolution System:
A drug with slow dissolution rate will yield an inherently sustained blood level. In principle, then it would seem possible to prepare controlled release products by controlling the dissolution rate of drugs that are highly water soluble. This can be done by preparing an appropriate salt or derivative, by coating the drug with a slowly soluble material, or by incorporating it into a tablet with a slowly soluble carrier. Ideally, the surface area available for dissolution must remain constant to achieve a constant release rate.
So far we have looked at the transfer of drugs in solution in the GIT, through a membrane, into solution in the blood. However, many drugs are given in solid dosage forms, & therefore must dissolve before absorption can take place.
If absorption is slow relative to dissolution then all we are concerned with is absorption. However, if dissolution is the slow, rate determining step (the step controlling the overall rate) then factors affecting dissolution will control the overall process. This is a more common problem with drugs which have a low solubility (below 1g/100 ml) or which are given at a high dose, e.g. griseofulvin (Robinson JR et al, 2005).
2.5. Materials:
Table: 2.2. Materials used in project work.
| Serial no. | Name | Category | Source | Country of origin |
| 1 | Metformin HCl | Active Drug | Maneesh pharmaceuticals pvt. ltd | India |
| 2 | Comet 500 XR | Tablet (Market preparation) | Square pharmaceuticals ltd. | Bangladesh |
| 3 | Glucomet 500 XR | Tablet (Market preparation) | Aristopharma ltd. | Bangladesh |
Table: 2.3. List of Instruments & Equipments used in Experiment.
| Serial No. | Name | Model |
| 1 | UV- Visible Spectrophotometer | HACH Spectrophotometer. |
Model-DR/4000u 2Tablet Dissolution Tester PHARMA TEST
Model-DT 70 3Electric Balance Denver Instrument
Model-M-310 4PH MeterLIDA Instrument Model-PHS-25
Table: 2.4. List of Apparatus/Glass wares used in this Project.
| Serial No | Name | Serial No | Name |
| 1 | Plastic container (10 L) | 8 | Measuring flask (1000ml) |
| 2 | Test tubes | 9 | Beakers |
| 3 | Volumetric flask (50ml & 100ml) | 10 | Disposable syringes (10ml) |
| 4 | Volumetric pipette | 11 | Glass rod |
| 5 | Micropipette | 12 | Filter |
| 6 | Pipette | 13 | Spatula |
7Measuring cylinder (25ml, 50ml, 1000ml) 14Mortar & pastels
Table: 2.5. List of reagents & Solvents.
| Serial No. | Name | Source | Country of origin |
| 1 | Potassium dihydrogen ortho phosphate | Technopharma | Bangladesh |
| 2 | Sodium hydroxide | Merck | Germany |
| 3 | Concentrated HCl (37%) | Merck | Germany |
| 4 | Purified water | Research Laboratory | Bangladesh |
2.6.1.1. Purified Water:
Purified water in bulk is prepared by distillation or by any other suitable method from water that complies with the regulations on water intended for human consumption laid down by the competent authority.
2.6.1.2. Preparation of 0.1N HCl:
For preparing 7L 0.1N HCl, 69.02ml of 37% conc. HCl was taken in a plastic container & then purified water was added up to 7L.
2.6.1.3. Preparation of Phosphate buffer:
For preparing 9L phosphate buffer, 61.2g of potassium dihydrogen ortho phosphate (K H2 PO4) was taken in a plastic container, dissolved in & diluted with distilled water up to the mark & then NaOH solution was added for pH (6.8) adjust.
2.7. Methods of dissolution study:
The dissolution of metformin HCl XR tablets from market preparation was monitored using standard BP apparatus paddle equipment with the tablet positioned 3mm above the bottom & UV spectrometer, at 232 nm. The compendially recommended stirring speed of 100 rpm for the paddle apparatus was used. The temperature was maintained at 37±0.5°C throughout. The apparatus was within specification for all six positions with regard to shaft wobble, alignment, rotation speed, vibration & temperature. Firstly, 0.1N HCl (about 900ml) was taken into six vessels & for each run, 3 tablets of Square Pharma & 3 tablets of Aristo Pharma were placed into six vessels respectively for 2 hr. These tablets were tested in basic buffer for 3to 8 hrs. At every interval, 10ml samples were withdrawn from the dissolution medium & 10ml of fresh buffer solution was added to each glass vessel to compensate the volume loss. The process was performed for 8 hrs to get a simulated picture of the drug release in the in vivo condition. The amount of drug released from the samples was then calculated with the help of appropriate calibration curve constructed from reference standard.
2.8. Assay Method:
Drug content of the sample solution i.e., the quantity of drug released in the dissolution medium from the metformin HCl XR tablet was determined after appropriate dilution (where necessary) by spectrophotometric analysis using a UV-Visible spectrophotometer, at 232 nm. 0.1N HCl & Phosphate buffer were used as the blank solution. From each value of absorbance, the concentration of the corresponding sample solution was calculated by using the equation of the standard curve for metformin HCl & then the amount of drug released in each vessel was determined. The percentages of drug release from the tablets were then calculated & plotted against time.
2.9. Theory of standard curve preparation:
The greater the number of molecule capable of absorbing light of a given wave length. The greater will be the extent of light absorption. Furthermore, the more effective a molecule is in absorbing light of a given wave length, the greater will be the extent of light absorption. From these guiding ideas the following empirical expression, which is known as the Beer-Lambert law, may be formulated:
Log (Io/I) = ε C l for a given wave length.
Where,
Io=Intensity of the light incident upon the cell.
I= Intensity of the light leaving the sample cell.
C=Molar concentration of solute.
L=Length of sample cell (cm).
Ε=Molar absorptivity.
2.9.1. Preparation of standard curve of metformin HCl:
- In case of Acidic Media:
50mg pure metformin HCl powder was dissolved in 0.1N HCl & made the
volume 100ml. So the concentration was 500μg/ml. It is called stock solu-
tion.
2. Different concentration of 5μg/ml, 10μg/ml, 15μg/ml, 20μg/ml, 25μg/ml,
30μg/ml, 35μg/ml were done to prepare a standard curve.
- To prepare these concentrations 1, 2, 3, 4, 5, 6, & 7ml of stock solution were taken, & made the volume 100ml with 0.1N HCl.
- The absorbance of these standard solutions of different concentration was measured at 232nm to construct the standard curve.
- In case of Basic Media:
50mg pure metformin HCl powder was dissolved in phosphate buffer &
made the volume 100ml. So the concentration was 500μg/ml. It is called
stock solution.
2. Different concentration of 2μg/ml, 4μg/ml, 6μg/ml, 8μg/ml, 10μg/ml
were done to prepare a standard curve.
- To prepare these concentrations 0.4, 0.8, 1.2, 1.6, & 2ml of stock solution were taken, & made the volume 100ml with phosphate buffer.
- The absorbance of these standard solutions of different concentration was measured at 232nm to construct the standard curve.
Table: 2.6. Absorbance of Metformin HCl for different concentration of standard solution (Acidic Media).
| Concentration (μg/ml) | Absorbance (232nm) |
| 0 | 0 |
| 5 | 0.092 |
| 10 | 0.175 |
| 15 | 0.261 |
| 20 | 0.351 |
| 25 | 0.435 |
| 30 | 0.525 |
| 35 | 0.611 |
Table: 2.7. Absorbance of Metformin HCl for different concentration of standard solution (Basic Media).
| Concentration (μg/ml) | Absorbance (232nm) |
| 0 | 0 |
| 2 | 0.171 |
| 4 | 0.341 |
| 6 | 0.501 |
| 8 | 0.671 |
| 10 | 0.832 |
3.0. Results & Discussion:
3.1. Results:
3.1.1. Zero order Plot:
Percentages (%) release of sample-1 (Square Pharmaceuticals ltd.) & sample-2 (Aristopharma) in acidic media (for 2hrs) & in basic media (for 3-8 hrs) are given below:
Table- 3.1: Percentages (%) of release of sample-1 (zero order).
| Time (hr) | Tablet-1 | Tablet-2 | Tablet-3 | Average % of release |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 18.5 | 19 | 21 | 19.5 |
| 2 | 30 | 35 | 28.5 | 31.16 |
| 4 | 37.25 | 40 | 45.25 | 40.83 |
| 6 | 61.20 | 68.75 | 67 | 65.65 |
| 8 | 80.27 | 78 | 82.83 | 80.36 |
Table-3.2: Percentages (%) of release of sample-2 (zero order).
| Time (hr) | Tablet-1 | Tablet-2 | Tablet-3 | Average % of release |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 25 | 17 | 30 | 24 |
| 2 | 45 | 31 | 50 | 42 |
| 4 | 48.9 | 67 | 61 | 58.96 |
| 6 | 68 | 79.23 | 78.88 | 75.37 |
| 8 | 87.5 | 95 | 90 | 90.83 |
3.1.2. Higuchi Plot:
Percentages (%) of release of sample-1 (Square Pharmaceuticals ltd.) & sample-2 (Aristopharma) in acidic media (for 2hrs) & in basic media (for 3-8 hrs) are given below:
Table- 3.3: Percentages (%) of release of sample-1 [Higuchi plot (SQRT)].
| Time (hr) | Tablet-1 | Tablet-2 | Tablet-3 | Average % of release |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 18.5 | 19 | 21 | 19.5 |
| 1.41 | 30 | 35 | 28.5 | 31.16 |
| 2 | 37.25 | 40 | 45.25 | 40.83 |
| 2.45 | 61.20 | 68.75 | 67 | 65.65 |
| 2.82 | 80.27 | 78 | 82.83 | 80.36 |
Table-3.4: Percentages (%) of release of sample-2 [Higuchi plot (SQRT)].
| Time (SQRT) | Tablet-1 | Tablet-2 | Tablet-3 | Average % of release |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 25 | 17 | 30 | 24 |
| 1.41 | 45 | 31 | 50 | 42 |
| 2 | 48.9 | 67 | 61 | 58.96 |
| 2.45 | 68 | 79.23 | 78.88 | 75.37 |
| 2.82 | 87.5 | 95 | 90 | 90.83 |
3.1.3. First order Plot:
Log percentages (%) remaining of sample-1 (Square Pharmaceuticals ltd.) & sample-2 (Aristopharma) in acidic media (for 2hrs) & in basic media (for 3-8 hrs) are given below:
Table- 3.5: Log percentages (%) remaining of sample-1 (First order).
| Time (hr) | Tablet-1 | Tablet-2 | Tablet-3 | Average Log % remaining of drug |
| 0 | 2 | 2 | 2 | 2 |
| 1 | 1.91 | 1.9 | 1.89 | 1.9 |
| 2 | 1.84 | 1.81 | 1.85 | 1.83 |
| 4 | 1.79 | 1.77 | 1.73 | 1.76 |
| 6 | 1.58 | 1.49 | 1.51 | 1.52 |
| 8 | 1.29 | 1.34 | 1.23 | 1.2 |
Table-3.6: Log percentages (%) remaining of sample-2 (First order)
| Time (hr) | Tablet-1 | Tablet-2 | Tablet-3 | Average Log % remaining of drug |
| 0 | 2 | 2 | 2 | 2 |
| 1 | 1.87 | 1.91 | 1.84 | 1.87 |
| 2 | 1.74 | 1.83 | 1.69 | 1.75 |
| 4 | 1.70 | 1.51 | 1.59 | 1.6 |
| 6 | 1.5 | 1.31 | 1.32 | 1.37 |
| 8 | 1.09 | 0.69 | 1.00 | 0.92 |
3.2. Discussion:
From the observation, we found out that release of drug from metformin HCl XR tablets was floating control system.
3.2.1. Comparative study of release kinetics from metformin HCl between sample-1 & sample-2 in acidic media:
Release kinetics of metformin HCl XR was studied using 0.1N HCl for 2hr. The release data were treated in different mathematical model for sample-1 & sample-2 to identify the release mechanism
Drug releases from sample-1 were 19.5% after 1hr, 31.16% after 2hr. On the other hand, drug releases from sample-2 were 24% after 1hr, 42% after 2hr
3.2.2. Comparative study of release kinetics from metformin HCl between sample-1 & sample-2 in basic buffer media:
Release kinetics of metformin HCl XR was studied using phosphate buffer (pH= 6.8) for 3-8 hr. The release data were treated in different mathematical model for sample-1 & sample-2 to identify the release mechanism
Drug releases from sample-1 were 40.83% after 4hr, 65.65% after 6hr, and 80.36% after 8hr. On the other hand, drug releases from sample-2 were 58.96% after 4hr, 75.37% after 6hr, and 90.83% after 8hr
3.2.3. Linearity observation between sample-1 & sample-2:
At zero order, from R2 values of sample-1 & sample-2, it was revealed that the release of drug from Metformin HCl XR tablets of sample-1 was more nearer to linearity (R2= 0.9751) than sample-2 (R2= 0.9505).
At Higuchi order plot, from R2 values of sample-1 & sample-2, it was revealed that the release of drug from Metformin HCl XR tablets of sample-2 was more nearer to linearity (R2= 0.991) than sample-1 (R2= 0.9556).
At first order plot, R2 values for sample-1 was 0.9671 that was more nearer to linear than sample-2 (R2= 0.9639), but that were not showed enough linearity as well as Higuchi order plots.
Finally, from the above discussion, we can say that sample-2 (Aristopharma) was better preparation than sampe-1 (Square pharma).
Conclusion
Metformin HCl is required 500 mg, 2 or 3 times daily in conventional dosage form. Sometimes the number and the frequency of doses are required to increase considering disease state. Repeated medication via rapid absorption causes side effects and toxicities along patient non-compliance and nocturnal harassment can be avoided by administering metformin HCl extended release dosage form, preferably once daily.
The influence of dissolution media on the release rate was significant found to be highest in phosphate buffer than in acidic media. The release data were then treated in different mathematical model to identify the release mechanism.
The approach of the present study was to make a comparative evaluation among the percent of drug release of two sample of metformin HCl XR tablets from two different companies. The study reveals the release of metformin HCl (water soluble drug) in acidic & basic media. The data generated in this study also shows that, the release pattern of drug closer to zero order & Higuchi release mechanism.
From the above discussion, the experiment indicates that, metformin HCl XR tablet delivered drug at desired rate. The rate and extent of drug release of different companies were different but was fully meet the BP specification.