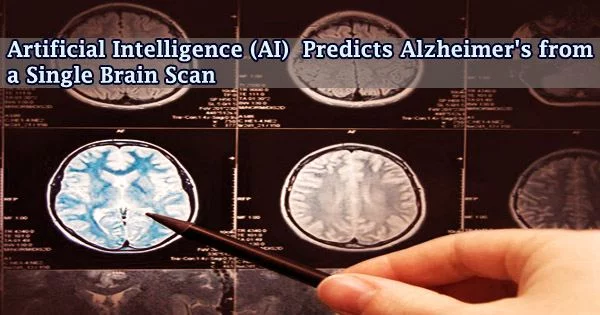
The most prevalent form of dementia is Alzheimer’s disease (AD), a neurodegenerative condition that gradually kills brain cells. A recent study demonstrates the use of artificial intelligence (AI) machine learning to create a biomarker for Alzheimer’s disease based on an MRI scan.
“In this study, we develop a method that uses magnetic resonance imaging data to identify differences in the brain between people with and without Alzheimer’s disease, including before obvious shrinkage of the brain occurs,” wrote the study authors. “This method could be used to help diagnose patients with Alzheimer’s Disease.”
According to the Alzheimer’s Association, there are currently 47 million people living with Alzheimer’s disease worldwide, and by 2030, that number is projected to rise to 76 million. According to a survey by AARP and the Women’s Alzheimer’s Movement (WAM), there are 5.8 million people with Alzheimer’s disease in the United States alone, and two-thirds of them are women.
According to the Harvard NeuroDiscovery Center at Harvard Medical School, there will be 16 million Americans with Alzheimer’s disease by the year 2050. Although Alzheimer’s disease has no known cure, there are therapies that could reduce some of the symptoms.
In this study, we develop a method that uses magnetic resonance imaging data to identify differences in the brain between people with and without Alzheimer’s disease, including before obvious shrinkage of the brain occurs. This method could be used to help diagnose patients with Alzheimer’s Disease.
Cami Rosso
A patient has more time to seek treatment to control their symptoms and receive assistance the earlier in the disease process they are diagnosed. Depending on the disease’s stage, Alzheimer’s symptoms can change. Short-term memory loss is typical in the early stages of the illness.
The capacity to carry out multiple tasks becomes deteriorated over time, which has an impact on executive functioning, motivation, focus, decision-making, problem-solving, judgment, and the capacity to multitask. People with AD lose the ability to carry out routine everyday duties as the disease progresses, and they eventually need carers to survive.
Depression, social disengagement, psychosis, wandering, apathy, agitation, suspicion of others, disinhibition, and delusions are a few of the neuropsychiatric symptoms of Alzheimer’s disease.
“In these last 40 years, improved computational power and storage capacity have led to numerous advances in developing non-invasive and low-cost structural biomarkers for AD that combine neuroimaging approaches, in particular structural MRI, with machine learning,” the researchers wrote.
The databases used for this study include the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database, the Open Access Series of Imaging Studied (OASIS) consortium, and the Imperial Memory Centre (IMC) based in London, United Kingdom.
The 115 sub-regions of magnetic resonance imaging scans included 45 regions in the white matter of the brain and 70 regions in the subcortical regions. Each region received over 650 unique features, including wavelet, intensity, texture, shape, and size, for evaluation.
The researchers modified a cancer-prediction AI classifier algorithm and trained it to recognize changes in brain characteristics that can indicate the existence of Alzheimer’s disease.
The algorithm was then tested on brain scan data from more than 80 patients at Imperial College Healthcare NHS Trust who were being assessed for AD, as well as data from the Alzheimer’s Disease Neuroimaging Initiative from more than 400 early and late-stage Alzheimer’s disease patients, a control group of people without Alzheimer’s disease, and patients with other brain disorders like Parkinson’s disease and front temporal dementia.
This work is unique in that the authors claim that the AI algorithm may be implemented into clinical decision support systems without extra patient testing or sample because it performed with 98 percent accuracy in its predictions.
Additionally, the AI technology was able to spot alterations in parts of the brain that hadn’t previously been linked to Alzheimer’s.
“Interestingly, the algorithm also selects regions not commonly related to AD, such as the cerebellum and the ventral diencephalon,” the scientists reported. “Together with a few studies reported in the literature, this outcome challenges the traditional view that white matter bundles in the cerebellum or in the ventral diencephalon are not affected by AD, possibly highlighting new therapeutic opportunities.”