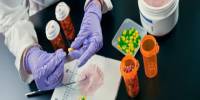
The production of chemicals needs to be done in a greener way. In a recent study, scientists from the University of Wisconsin-Madison present one possible route toward achieving this objective by modifying hydrogen fuel cell technologies. Some electric cars, laptops, and cell phones are already powered by these technology.
“The chemical industry is a massive energy consumer, and there is a big push to decarbonize the industry,” says Shannon Stahl, a professor in the UW-Madison Department of Chemistry who guided much of the research. “Renewable electricity can provide energy to produce chemicals with a much lower carbon footprint than burning fossil fuels.”
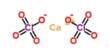
The traditional method employs a lot of zinc metal as an electron source, but processing zinc is difficult and produces a lot of trash that is bad for the environment. Working with scientists at the pharmaceutical maker Merck & Co. Inc., UW-Madison chemists and engineers sought to develop a more sustainable method to manufacture ingredients needed to make many types of drugs.
In their search for an alternative process, the researchers took inspiration from hydrogen fuel cells, which use hydrogen gas as the source of electrons to generate electricity.
“The process we are working with needs a green source of electrons,” says Stahl. “We realized that fuel cell technology could be modified to make chemicals rather than electricity.”
This work is connected to a broader effort to create a hydrogen infrastructure that goes beyond fuel cells and energy production. This work shows that hydrogen can be combined with electricity to make new drugs.
Mathew Johnson
Hydrogen gas is an ideal choice in many ways, according to Stahl. It can be generated from renewable electricity, and it creates very little waste. Developing a hydrogen-based way to make pharmaceuticals aligns with renewed interest in a “hydrogen economy.”
“This work is connected to a broader effort to create a hydrogen infrastructure that goes beyond fuel cells and energy production,” says Mathew Johnson, a postdoctoral researcher in the chemistry department who led the study. “This work shows that hydrogen can be combined with electricity to make new drugs.”
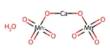
The scientists created a mechanism that draws electrons away from hydrogen using a type of organic chemical called a quinone. The fact that this method functions well without water is a crucial aspect. Water is normally required for fuel cells to function properly, however water can interfere with processes that are used to create medicinal components.
The system then uses electricity to supercharge the electrons, giving the electrons more energy than hydrogen could normally provide.
The team, which included postdoctoral researcher Jack Twilton, chemistry professor Daniel Weix and chemical and biological engineering professor Thatcher Root, described their new system in a paper published August 21, 2023, in the journal Nature. They show how it can be used to make dozens of important organic molecules, including a large batch of a pharmaceutical ingredient.
The group is currently focusing on enhancing the procedure to enable industrial-scale production. And Stahl and his collaborators see even bigger opportunities for this technology.
“This is a broadly applicable technology for chemical production,” says Johnson. “Many chemical processes need electrons. This is not limited to pharmaceuticals. It should be a very versatile technology.”