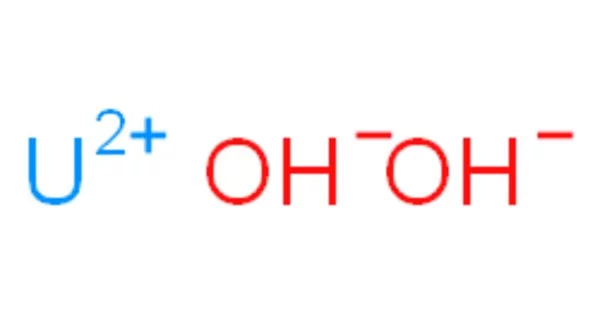
Uranyl hydroxide is a uranium hydroxide with the chemical formula UO2(OH)2 in monomeric form and (UO2)2(OH)4 in dimeric form, both of which can exist in normal aqueous media. Uranyl hydroxide hydrate precipitates as a colloidal yellowcake from oxidized uranium liquors with a pH close to neutral.
It is a chemical compound made up of one uranium atom and six fluorine atoms. It is the chemical form of uranium used in the enrichment of uranium. It can be a solid, liquid, or gas within a reasonable range of temperature and pressure. Solid UF6 is a white, dense, crystalline substance that is similar to rock salt.
Uranyl hydroxide is teratogenic and radioactive.
Properties
- Chemical formula: UH2O4
- Molar mass: 304.0424 g mol−1
- Related compounds: Sulfuric acid, Chromic acid, Uranium trioxide
Reaction
Uranium Hydroxide does not react with oxygen, nitrogen, carbon dioxide, or dry air, but it does react with water or water vapor. As a result, UF6 is always handled in leak-proof containers and processing equipment. When UF6 comes into contact with water, such as water vapor in the air, the UF6 and water react, producing corrosive hydrogen fluoride (HF) and a uranium-fluoride compound known as uranyl fluoride (UO2F2).
The gaseous diffusion process used to enrich uranium requires uranium in the form of UF6. In the first step of UF6 production, uranium ore is mined and transported to a mill where uranium oxide (also known as “yellowcake”) is produced. The uranium oxide is then transported to a UF6 production facility. At the production facility, the uranium oxide is combined with anhydrous HF and fluorine gas in a series of chemical reactions to form the chemical compound UF6. The product UF6 is placed into steel cylinders and shipped as a solid to a gaseous diffusion plant for enrichment.
Application
Uranyl hydroxide was previously used in glassmaking and ceramics to color vitreous phases and to prepare pigments for high temperature firing. When alkaline diuranates (such as sodium diuranate) are introduced into glasses, they produce yellow by transmission and green by reflection; additionally, these glasses become dichroic and fluorescent when exposed to ultraviolet rays.
It is used in uranium processing because of its distinct properties, which make it extremely convenient. It can be used as a gas for processing, a liquid for filling or emptying containers or equipment, and a solid for storage, all at temperatures and pressures typical of industrial processes.