A nanopore is a pore with a diameter of one nanometer. It could be formed by a pore-forming protein or as a hole in synthetic materials like silicon or graphene. A nanopore in an electrically insulating membrane can serve as a single-molecule detector. A biological protein channel in a high electrical resistance lipid bilayer, a pore in a solid-state membrane, or a hybrid of these – a protein channel embedded in a synthetic membrane – are all possibilities.
The detection principle involves measuring the ionic current flowing through the nanopore as a voltage is applied across the membrane. When the nanopore has molecular dimensions, molecules (e.g., DNA) passing through cause interruptions in the “open” current level, resulting in a “translocation event” signal. The passage of RNA or single-stranded DNA molecules, for example, causes a ~90% blockage of the current through the membrane-embedded alpha-hemolysin channel (1.5 nm diameter) (measured at 1 M KCl solution).
Researchers have discovered that the conditions inside tiny pores can have a significant impact on chemistry. Many natural and engineered materials contain an entire aqueous universe hidden within their tiny pores. According to research from Washington University in St. Louis’ McKelvey School of Engineering, when such materials are submerged in liquid, the chemistry inside the tiny pores – known as nanopores – can differ significantly from that in the bulk solution.
pH is a ‘master variable’ for water chemistry. When it is measured in practice, people are really measuring the pH of the bulk solution, not the pH inside the material’s nanopores. And if they are different, that is a big deal because the information about the little tiny space will change the entire prediction in the system.
Young-Shin Jun
In fact, in higher-salinity solutions, the pH inside of nanopores can be as much as 100 times more acidic than in the bulk solution. The research findings were published in the journal CHEM.
A better understanding of nanopores can have significant implications for many engineering processes. Consider membrane processes for producing clean water, decarbonization technologies for energy systems, such as carbon capture and sequestration, hydrogen production and storage, and batteries.
Young-Shin Jun, a professor of energy, environmental, and chemical engineering, and Srikanth Singamaneni, the Lilyan & E. Lisle Hughes Professor in the Department of Mechanical Engineering & Materials Science, wanted to know how the pH of nanopores differed from the bulk liquid solution they were submerged in.
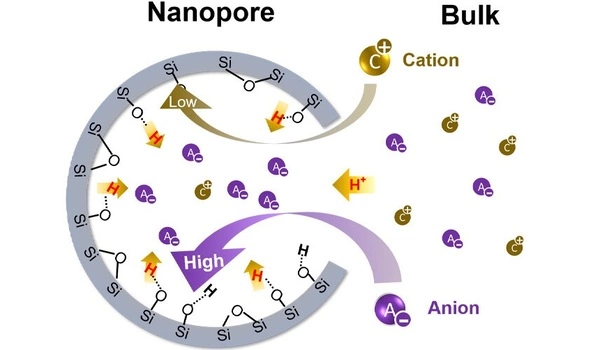
“pH is a ‘master variable’ for water chemistry,” Jun said. “When it is measured in practice, people are really measuring the pH of the bulk solution, not the pH inside the material’s nanopores. And if they are different, that is a big deal because the information about the little tiny space will change the entire prediction in the system.”
Aside from rapid DNA sequencing, other applications include the separation of single and double stranded DNA in solution and the determination of polymer length. At this point, nanopores are contributing to our understanding of polymer biophysics, single-molecule analysis of DNA-protein interactions, and peptide sequencing.
When it comes to peptide sequencing, bacterial nanopores such as hemolysin can be used for RNA, DNA, and, more recently, protein sequencing. For example, in a study in which peptides with the same Glycine-Proline-Proline repeat were synthesized and then put through nanopore analysis, an accurate sequence was obtained.