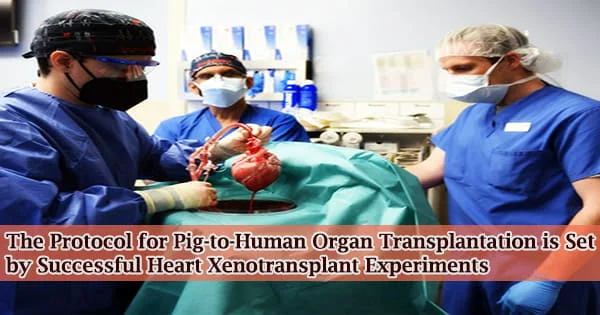
Two genetically modified pig hearts were successfully transplanted into recently deceased humans in June and July by a team at NYU Langone Health, marking the most recent developments in the fight against the national organ shortage and the creation of a clinical protocol that would provide an alternative source of organs for people with life-threatening heart disease.
The procedures, known as xenotransplant, were carried out at NYU Langone’s Tisch Hospital on Thursday, June 16, 2022, and Wednesday, July 6, 2022. The investigational procedures were carried out under the direction of Nader Moazami, MD, surgical director of heart transplantation at the NYU Langone Transplant Institute, using hearts obtained from a facility hundreds of miles away and transplanted into recently deceased donors kept on ventilator support.
The heart function was monitored for three days while the transplant surgeries took place over a period of several hours. On Sunday, June 19, 2022, the first heart xenotransplant was completed, and on July 9, 2022, the second.
The hearts functioned normally with the usual post-transplant drugs and without further mechanical support, and neither organ showed any early signs of rejection. In neither case was porcine cytomegalovirus (pCMV) found using a novel infectious disease technique.
Strict procedures were also followed to prevent and track potential zoonotic transmission of the porcine endogenous retrovirus (PERV). In order to be exclusively used for xenotransplantation research moving forward, the operating room used for this study has been taken offline.
The hearts came from pigs with 10 genetic modifications, including 6 human transgenes (“knock-ins”) to promote the expression of proteins that regulate critical biologic pathways that can be disrupted by incompatibilities between pigs and humans and 4 porcine genes “knockouts” to prevent rejection and abnormal organ growth.
Our goal is to integrate the practices used in a typical, everyday heart transplant, only with a nonhuman organ that will function normally without additional aid from untested devices or medicines. We seek to confirm that clinical trials can move ahead using this new supply of organs with the tried-and-true transplant practices we have perfected at the NYU Langone Transplant Institute.
Dr. Nader Moazami
In this NYU Langone Health trial, no further investigative tools or drugs were used. Current clinical standards utilized in heart transplantation were followed for the procurement, transport, transplant operation, and immunosuppression.
“Our goal is to integrate the practices used in a typical, everyday heart transplant, only with a nonhuman organ that will function normally without additional aid from untested devices or medicines,” said Dr. Moazami. “We seek to confirm that clinical trials can move ahead using this new supply of organs with the tried-and-true transplant practices we have perfected at the NYU Langone Transplant Institute.”
These most recent developments in xenotransplantation, according to Alex Reyentovich, MD, medical director of heart transplantation and director of the NYU Langone Advanced Heart Failure program, get the field closer to achieving a fresh supply of organs for individuals facing a life-threatening disease.
“These are the first steps in developing a deep understanding of the mechanical, molecular, and immunologic aspects of xenoheart transplantation and the feasibility of utilizing standard clinical practice and tools to do so,” said Dr. Reyentovich.
Decedent Model Key to Xenotransplant Research
Robert Montgomery, MD, DPhil, the H. Leon Pachter, MD, Professor and chair of the Department of Surgery at NYU Grossman School of Medicine and director of the NYU Langone Transplant Institute, said xenotransplant studies with recently deceased donors are critical to gathering the additional human data needed to advance a field that for decades until last year had been tested only using nonhuman primates.
“Our greater purpose is to address the organ shortage and provide another option for the more than 100,000 people nationwide waiting on that lifesaving gift,” he said. “The paradigm of whole-body donation when organ donation is not a viable option is critical to this work moving forward. We are so grateful to the families who volunteer to participate in this research, which will lead to saving untold thousands of more lives.”
The operations directed by Dr. Moazami were carried out after conferring with the New York State Department of Health as part of a wider study that was approved by a specially designated research oversight board at NYU Langone. It is the most recent action in a study protocol that requires further and comparable actions to be taken.
When a person has been declared brain dead and their organs or tissues cannot be used for transplantation, a new pathway that enables them to realize their altruism is whole-body donation after death for the benefit of groundbreaking investigations.
“As the organ procurement organization for the greater New York area we are supportive of NYU Langone’s research efforts,” said Leonard Achan, RN, MA, ANP, president and chief executive officer at LiveOnNY.
“Under normal circumstances, one donor can save up to eight lives. But in these special cases, donor heroes have the potential to save countless lives by participating in groundbreaking research. We can never thank these donor heroes and their families enough for saying yes and helping others. It is critically important to continue advancing this research.”
Enhancing Infectious Disease Protocol
Dr. Montgomery, who oversaw ground-breaking xenotransplant surgeries using single-gene knockout pig kidneys in September and November 2021, says improved porcine virus monitoring, which was incorporated in these most recent procedures, is one of the critical elements of success to advancing this field.
“Other studies have shown that pCMV may be a factor in the success of xenotransplanted organs,” he said. “More sensitive screening methods have been introduced to detect low-level traces of pCMV in the donor pigs. We have included that additional screening in this heart transplant protocol to give the organ the best chance at long-term survival.”
Sapna Mehta, MD, director of the transplant infectious diseases program and clinical director of the NYU Langone Transplant Institute, was in charge of developing the infectious disease testing methodology at NYU Langone Health.
Hearts from pigs created by Revivicor, Inc., a division of United Therapeutics Corporation, which provided funding for this study, were used in these procedures.