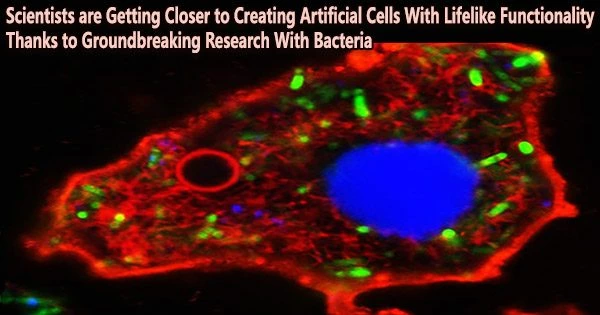
Researchers have used bacteria to create sophisticated synthetic cells that imitate the activity of genuine cells.
The study, led by the University of Bristol and published today in Nature, advances the use of artificial cells, or protocells, to more faithfully mimic the intricate makeup, structure, and function of living cells.
Achieving true-to-life functioning in protocells is an international great challenge involving several disciplines, from beginning of life research to bottom-up synthetic biology and bioengineering.
Due to previous failures in modeling protocells using microcapsules, the study team turned to bacteria to construct sophisticated synthetic cells utilizing a living material assembly method.
Professor Stephen Mann from the University of Bristol’s School of Chemistry, and the Max Planck Bristol Centre for Minimal Biologytogether with colleagues Drs Can Xu, Nicolas Martin (currently at the University of Bordeaux) and Mei Li in the Bristol Centre for Protolife Research have demonstrated an approach to the construction of highly complex protocells using viscous micro-droplets filled with living bacteria as a microscopic building site.
The group first exposed the empty droplets to two different bacterial species. Although the other population was imprisoned at the droplet surface, one population spontaneously became stuck inside the droplets.
The liberated cellular components were then retained inside or on the surface of the droplets by the destruction of both types of bacteria, resulting in membrane-coated bacteriogenic protocells that contained hundreds of biological molecules, parts, and machinery.
Our living-material assembly approach provides an opportunity for the bottom-up construction of symbiotic living/synthetic cell constructs. For example, using engineered bacteria it should be possible to fabricate complex modules for development in diagnostic and therapeutic areas of synthetic biology as well as in biomanufacturing and biotechnology in general.
Dr. Can Xu
The fact that the protocells could generate RNA and proteins by in vitro gene expression as well as energy-rich molecules (ATP) via glycolysis suggested that the hereditary bacterial components persisted in the synthetic cells.
The scientists used a number of chemical processes to physically and morphologically alter the bacteriogenic protocells in order to test the technique’s effectiveness further. The interior of the droplet was filled with a cytoskeletal-like network of protein filaments and membrane-bound water vacuoles, and the liberated bacterial DNA was compressed into a single structure resembling a nucleus.
As a step towards the building of a synthetic/living cell entity, the researchers injected living bacteria into the protocells to provide self-sustainable ATP synthesis and long-term energization for glycolysis, gene expression and cytoskeletal assembly.
Interestingly, the protoliving constructions developed an exterior appearance resembling an amoeba as a result of local bacterial growth and metabolism, creating a cellular bionic system with integrated life-like features.
Corresponding author Professor Stephen Mann said: “Achieving high organisational and functional complexity in synthetic cells is difficult especially under close-to-equilibrium conditions. Hopefully, our current bacteriogenic approach will help to increase the complexity of current protocell models, facilitate the integration of myriad biological components and enable the development of energised cytomimetic systems.”
First author Dr. Can Xu, Research Associate at the University of Bristol, added: “Our living-material assembly approach provides an opportunity for the bottom-up construction of symbiotic living/synthetic cell constructs. For example, using engineered bacteria it should be possible to fabricate complex modules for development in diagnostic and therapeutic areas of synthetic biology as well as in biomanufacturing and biotechnology in general.”