Researchers from Duke Health have discovered the underlying cause of a hereditary metabolic disorder that affects people of Lumbee and other Native American ancestry often, challenging decades of received wisdom and opening the door to novel, more potent treatments.
The discovery, which appeared published on April 19, 2023, in the journal Science Translational Medicine, challenges textbook theories for how a certain protein degrades in a child’s brain, becomes toxic, and causes potentially deadly neurological issues.
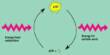
The inherited condition is called Glutaric Aciduria Type I (GA-1), and current literature describes the toxic substances as being produced in the brain instead of arising elsewhere and crossing the blood-brain barrier.
Treatments for the condition, including a strict, low-protein diet, have limited success. Up to one-third of affected children experience long-term brain impairment, and some of them pass away.
The Duke researchers reopened the research into GA-1 because it has been demonstrated that other metabolic illnesses can affect the brain by causing the liver to breakdown proteins. Senior author Karl-Dimiter Bissig, M.D., Ph.D., an associate professor in Duke’s departments of Pediatrics, Medicine, Biomedical Engineering and Pharmacology and Cancer Biology, led the work.
Bissig and colleagues launched experiments in mice specially bred to have GA-1. They discovered that catabolites residue left over from the breakdown of lysine, an important amino acid aggregate in the liver and can pass through the blood-brain barrier. As a result, there is a toxic buildup of glutaric acid in the brain, which damages the nerves and affects motor function.
The original experiments led to the interpretation that the toxic catabolites were produced locally in the brain. What our work demonstrates is the importance of challenging paradigms, particularly as new technologies and research approaches are available.
Karl-Dimiter Bissig
Either a liver donation or CRISPR gene-editing technology allowed the researchers to successfully treat the disease in mice. Other gene therapies that target the liver may also be beneficial and only need to be used once in a lifetime.
“The original experiments led to the interpretation that the toxic catabolites were produced locally in the brain,” Bissig said. “What our work demonstrates is the importance of challenging paradigms, particularly as new technologies and research approaches are available.”
Bissig said inadequate measures to address different mutations in specific populations are leading to health disparities. People with Native American, Amish and Irish heritage have high susceptibility to GA-1, which can be identified during newborn screenings; the genetic variant common in Lumbee populations seems to cause the most damaging disease.
Since states choose which illnesses should be screened for in newborn screenings, GA-1 is not detected if it is not on the state’s selected screening panel. Screenings could also be missed if babies are delivered at home.
Although early detection and a low-protein diet have saved lives, the advantages are concentrated among children of Amish and Irish ancestry, who traditionally have had better access to healthcare than Native Americans.
“With a better understanding of this disease, we can now work to develop treatments that are more effective and easier to access,” Bissig said. “It’s much easier to treat the liver than the brain. We are now working to advance the more efficient and convenient therapies.”