Magnetic resonance imaging (MRI) scans use gadolinium contrast media, which are chemical substances. Gadolinium contrast medium, when injected into the body, enhances and improves the quality of MRI images (or pictures). This enables the radiologist to provide more accurate information about how your body is functioning and whether there is any disease or abnormality present.
The magnetic interactions between gadolinium contrast agents used in MRI scans and their environment are revealed by simulations. You are free to keep your best guesses. Engineers at Rice University’s George R. Brown School of Engineering are learning more about what happens when contrast agents are injected into your body for an MRI scan.
In a new study that could lead to better scans, a Rice-led team digs deeper via molecular simulations that, unlike earlier models, make absolutely no assumptions about the basic mechanisms at play when gadolinium agents are used to highlighting soft tissues.
The study led by Rice chemical and biomolecular engineer Philip Singer, former associate research professor Dilip Asthagiri, now of Oak Ridge National Laboratory, and graduate student Thiago Pinheiro dos Santos appears in Physical Chemistry Chemical Physics.
Overall, these findings open up a new avenue for understanding how contrast agents respond to human body conditions during an MRI scan. By better understanding this, one can create new, safer, and more sensitive contrast agents, as well as use simulations, to improve MRI data interpretation.
Philip Singer
It uses sophisticated models developed at Rice for oil and gas studies to definitively analyze how hydrogen nuclei at body temperatures “relax” under nuclear magnetic resonance (NMR), the technology used by magnetic resonance imaging, also known as MRI.
Doctors use MRI to “see” the state of a patient’s soft tissues, including the brain, by causing magnetic moments in the hydrogen nuclei of water molecules to align with the magnetic field, a process that can be manipulated when gadolinium agents are present. When the aligned nuclei relax back to thermal equilibrium after excitation, the device detects bright spots. The brighter the contrast, the faster they relax.
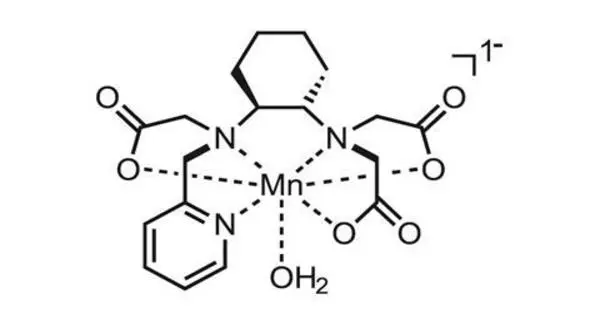
Gadolinium molecules are paramagnetic by nature and are sensitive to magnetic excitation. They are usually chelated as part of a contrast agent because they are toxic. “Basically, a chelate hugs the gadolinium and protects your body from directly interacting with the metal,” Pinheiro dos Santos explained. “We want to know how these molecules behave.”
Despite the fact that gadolinium-based contrast agents are injected into patients by the ton every year, their molecular mechanism has never been fully understood.
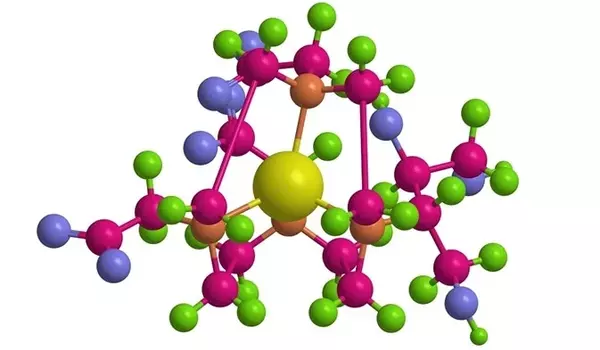
“Going back 40 years, in the NMR field people assumed liquid water is just a collection of marbles moving about, and the dipoles in the marbles randomly reorient,” Asthagiri said. But such assumptions are limiting, he said. “What Thiago does with his explicit simulation is show how the water network evolves in time,” Asthagiri said. “These are complicated, computationally intensive calculations.”
To study the phenomenon in depth, the Rice simulations used highly refined, polarizable force fields, which necessitated intensive GPU-accelerated computing. Co-author Steven Greenbaum, a professor of physics at Hunter College in the City University of New York, whose lab specializes in NMR measurements of ionic and molecular transport processes in condensed matter, validated the team’s molecular dynamics approach with experimental data.
The simulations revealed significant differences in the responses of the inner and outer shells of water molecules surrounding gadolinium to thermal excitation. “The inner shell is made up of eight or nine water molecules that surround gadolinium,” Pinheiro dos Santos explained. “They’re strongly bound to the gadolinium and remain there for a long time, a few nanoseconds. The outer shell encompasses all of the remaining water molecules.”
The researchers discovered that, while the structure of the inner shell remains constant between 41 and 98.6 degrees Fahrenheit, its dynamics are extremely sensitive to thermal effects. They also discovered that temperature has a significant impact on the self-diffusivity of molecules in gadolinium-water simulations, which affects outer-shell relaxation.
“Overall, these findings open up a new avenue for understanding how contrast agents respond to human body conditions during an MRI scan,” Singer said. “By better understanding this, one can create new, safer, and more sensitive contrast agents, as well as use simulations to improve MRI data interpretation.”
He said future studies will examine chelated gadolinium complexes in fluids that are more representative of cellular interiors.