Physical activity is beneficial to a mouse’s brain, as well as yours. This has been proven in numerous investigations involving mice, humans, and laboratory glassware. A new study demonstrates that the cognitive benefits obtained by marathon-running mice may be transferred to their couch-potato counterparts.
Blood from young adult mice who get a lot of activity enhances the brains of the same-aged, sedentary mice, according to Stanford School of Medicine researchers. This benefit appears to be mostly due to a particular protein found in the blood of exercising mice.
The discovery could pave the way for medicines that reduce the risk of neurodegenerative illness or halt its course in people who don’t get enough exercise.
The Stanford researchers examined blood samples from athletic and sedentary mice of the same age in their study, which will be published in Nature on Dec. 8. They discovered that giving sedentary mice blood from sprinting mice reduced neuroinflammation and increased cognitive performance.
Furthermore, the researchers discovered a blood-borne protein that appears to be crucial in the anti-inflammatory exercise effect.
Inflammation and cognitive health
According to Tony Wyss-Coray, Ph.D., professor of neurology and neurological sciences, neuroinflammation has been closely linked to neurodegenerative illnesses in humans. According to him, animal studies have shown that neuroinflammation causes neurodegenerative illnesses and that reversing or lowering neuroinflammation can help people live longer.
Anybody who’s suffered from influenza can relate to the loss of cognitive function that comes from a fever-inducing viral infection, Wyss-Coray said: “You get lethargic, you feel disconnected, your brain doesn’t work so well, you don’t remember as clearly.”
This is due, at least in part, to the infection’s resulting bodywide inflammation. The inflammation in your brain spreads as your immune system cranks up its assault. According to Wyss-Coray, a neuro-immunologist who detected indications of brain inflammation in persons who died of COVID-19 in a study published earlier this year, neuroinflammation accelerates the onset of Alzheimer’s and other neurodegenerative disorders.
The runners’ blood was clearly doing something to the brain, even though it had been delivered outside the brain, systemically.
Wyss-Coray
The new study’s senior author is Wyss-Coray. Zurine De Miguel, Ph.D., a former Wyss-Coray postdoctoral scholar who is currently an assistant professor of psychology at California State University, Monterey Bay, is the lead author.
Exercise is already known to provide a lot of beneficial effects in the brain, such as increased nerve cell development and reduced inflammation.
“We’ve discovered that this exercise effect can be attributed to a large extent to factors in the blood, and we can transfer that effect to a same-aged, non-exercising individual,” said Wyss-Coray, the D. H. Chen Professor II.
Nightly mouse marathon
Mice enjoy running. Give confined mice access to a few-inch-diameter running wheel, and it will run 4 to 6 miles per night (they sleep during the day) on legs that are considerably shorter than ours without any training or prodding.
The mouse won’t get nearly as much exercise if you lock the wheel, but it’ll still be allowed to scurry around its cage (roughly equivalent to heading into the kitchen now and then to fetch a beer or a snack from the fridge).
The researchers placed either working or locked running wheels in the cages of 3-month-old lab mice, which have a metabolic age of 25 years. When marathoner mice were compared to inactive mice, a month of consistent running was enough to significantly increase the number of neurons and other cells in their brains.
The researchers next took blood samples from marathon runners and inactive mice as controls. They next injected plasma (the cell-free part of the blood) from marathoner or couch-potato animals into additional inactive mice every three days.
Each injection accounted for 7% to 8% of the total blood volume of the recipient mouse. (An equivalent amount in humans would be about ½ to ¾ of a pint.)
“The mice getting runner blood were smarter,” Wyss-Coray said.
Sedentary mice treated with marathoner plasma beat their equally sedentary friends that got couch-potato plasma in two memory tests.
Furthermore, sedentary mice given marathoner mice’s plasma had more cells that give rise to new neurons in the hippocampus (a brain region related with memory and navigation) than that given couch-potato plasma.
The researchers evaluated the levels of activation of hundreds of genes in the brain of sedentary animals given marathoner vs couch-potato plasma. The activation levels of the 250 genes whose activation levels changed the greatest in response to marathoner plasma were known to be most strongly associated to inflammatory processes, and their activation-level alterations showed decreased neuroinflammation in mice given marathoner blood transfusions.
“The runners’ blood was clearly doing something to the brain, even though it had been delivered outside the brain, systemically,” said Wyss-Coray.
When the Stanford team looked at the proteins in the marathoner mice’s blood, they found 235 different proteins, 23 of which were scarcer and 26 more abundant in marathoner mice than in couch-potato mice.
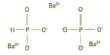
Several of the differently elevated proteins were linked to the complement cascade, which is a collection of roughly 30 blood-borne proteins that interact to kick-start the immune response to infections.
Chronic inflammation caused by abnormal complement activation appears to hasten the progression of several neurodegenerative diseases, according to Wyss-Coray.
A protein of interest
The anti-inflammatory impact of clusterin, a single protein found in marathoner mice’s plasma, was largely nullified when it was removed from sedentary mice’s brains. No other protein examined in the same way got the same result.
Clusterin, a complement cascade inhibitor, was found in much higher concentrations in marathoners’ blood than in couch potatoes’ blood.
Clusterin attaches to receptors found on brain endothelial cells, the cells that line the brain’s blood vessels, according to further research. The majority of Alzheimer’s patients have inflamed blood endothelial cells, according to Wyss-Coray, whose research has revealed that blood endothelial cells may transmit chemical signals from circulating blood into the brain, including inflammatory signals.
Clusterin, when injected outside the brain, was able to diminish brain inflammation in two separate strains of lab mice that had been provoked with either acute body-wide inflammation or Alzheimer’s-related chronic neuroinflammation.
Separately, the researchers discovered that 20 military veterans with moderate cognitive impairment, a precursor to Alzheimer’s disease, had raised clusterin levels in their blood after completing a six-month aerobic exercise program.
According to Wyss-Coray, a medication that boosts or mimics clusterin’s binding to its receptors on brain endothelial cells could help reduce the progression of neuroinflammation-related neurodegenerative illnesses like Alzheimer’s.
Wyss-Coray is a faculty fellow at Stanford ChEM-H and a member of the Stanford Wu Tsai Neuroscience Institute, Stanford Bio-X, and the Stanford Maternal and Child Health Research Institute.
Other Stanford study co-authors are former postdoctoral scholars Nathalie Khoury, Ph.D., Niclas Olsson, Ph.D., Ryan Vest, Ph.D., and Hui Zhang, Ph.D.; former graduate student Michael Betley, DVM, Ph.D.; former neurology instructor Benoit Lehallier, Ph.D.; former undergraduate Drew Willoughby; postdoctoral scholars Andrew Yang, Ph.D., Oliver Hahn, Ph.D., and Nannan Lu, Ph.D.; former medical and graduate student Liana Bonanno, MD, Ph.D.; Palo Alto Veterans Institute for Research assistant Lakshmi Yerra; former staff scientist Lichao Zhang, Ph.D.; laboratory manager Nay Lui Saw; former life sciences research associate Davis Lee; Kaci Fairchild, Ph.D., clinical assistant professor of psychology; former life science research professional Patrick McAlpine; Mehrdad Shamloo, Ph.D., professor of neurosurgery; Joshua Elias, Ph.D., assistant professor of chemical and systems biology; and Thomas Rando, MD, Ph.D., professor of neurology and neurological sciences.
The study was funded by the National Institutes of Health (grants AG0047820 and 1F32AG067652), the Stanford Alzheimer’s Disease Research Center (NIH grant P30 AG066515), the U.S. Department of Veterans Affairs, the U.S. Department of Defense, the Alzheimer’s Association, and the Marie Curie Foundation, the NOMIS Foundation, the Simons Foundation and the Wu Tsai Neurosciences Institutes’ Brain Rejuvenation Project with support from the Bertarelli Foundation
Stanford’s Department of Neurology and Neurological Sciences also supported the work.









![Report on Primary School Dropouts The Reasons Behind an Anthropological Investigation [part-4]](https://assignmentpoint.com/wp-content/uploads/2013/04/images-8-110x55.jpg)