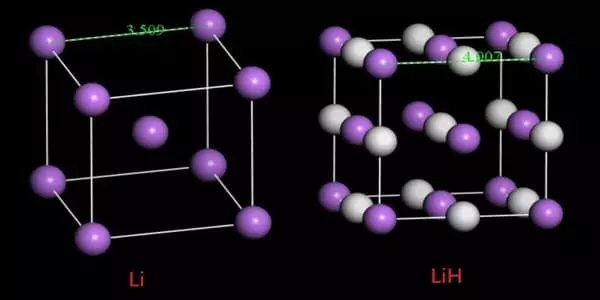
Lithium hydride, abbreviated LiH, is an inorganic compound. Although commercial samples are grey, this alkali metal hydride is a colorless solid. It is a gray crystalline solid formed by the direct combination of its constituent elements at high temperatures, and it is a ready source of hydrogen, releasing that gas instantly when treated with water. It is a salt-like (ionic) hydride with a high melting point that is insoluble but reactive with all protic organic solvents.
It is soluble and inert with certain molten salts, including lithium fluoride, lithium borohydride, and sodium hydride. It is the lightest ionic compound, with a molecular mass of slightly less than 8.0.
Properties
Lithium hydride appears as a white or translucent crystalline mass or powder. The commercial product is light bluish-gray lumps due to the presence of minute amounts of colloidally dispersed lithium.
- Molar Mass/Weight: 7.948 g/mol
- Color and Appearance: Off-white, gray, or translucent; light bluish-gray lumps in commercial product
- Odor: Odorless
- Melting Point: 680 °C, 1256 °F
- Boiling Point: 900-1000 °C, 1650-1830 °F (decomposes)
- Density: 0.76-0.8 g cm-3
- State of matter at room temperature: Solid
Physical properties
LiH is a diamagnetic and ionic conductor with a conductivity that gradually increases from 2×10-5 Ω-1 cm-1 at 443°C to 0.18 Ω-1 cm-1 at 754°C with no discontinuity through the melting point. LiH’s dielectric constant falls from 13.0 (static, low frequencies) to 3.6. (visible-light frequencies). LiH is a soft material with a hardness of 3.5 on the Mohs scale. Its compressive creep (per 100 hours) rapidly increases from 1% at 350 °C to >100% at 475 °C, indicating that heated LiH cannot provide mechanical support.
The thermal conductivity of LiH decreases with temperature and morphology: at 50 °C, the corresponding values are 0.125 W/(cm•K) for crystals and 0.0695 W/(cm•K) for compacts, and at 500 °C, the corresponding values are 0.036 W/(cm•K) for crystals and 0.0432 W/(cm•K) for compacts. At room temperature, the linear thermal expansion coefficient is 4.2×105/°C.
How to make Lithium Hydride
LiH can be synthesized by the reaction of solid lithium with hydrogen gas, which is represented by the following chemical equation:
2Li + H2 → 2LiH
The reaction occurs rapidly at temperatures over 600 °C, and the yield is increased to 98%. It can also occur at 29 °C, but the yield is much lower.
Lithium hydride is also produced by combining lithium aluminum hydride and sodium hydroxide through metathesis in tetrahydrofuran (THF):
LiAlH4 + NaH → NaAlH4 + LiH
Reactions with Other Compounds
LiH reacts with air to produce Li2O, LiOH, and Li2CO3, while it forms nitrogenous compounds in moist air. It spontaneously undergoes a chemical reaction with water which is given as follows:
LiH + H2O → Li+ + H2 + OH–
Lithium hydride reacts with sulfur dioxide to form lithium dithionite at a temperature above 50 °C:
2LiH + 2SO2 → Li2S2O4 + H2
When it reacts with acetylene, it produces hydrogen and lithium carbide:
LiH + LiOH →Li2O + H2
Uses
Lithium hydride is a material that can be used to store hydrogen as well as a Li+ ion conductor. LiH is primarily used as a precursor in the synthesis of complex hydrides.
It is used in the production of lithium aluminum hydride and silane, as a strong reducing agent, as a condensation agent in organic synthesis, as a portable hydrogen source, and as a lightweight nuclear shielding material. It is now being used for thermal energy storage in space power systems.