Reduced mercury emissions from industry are an important step towards reducing the environmental and health impacts of the industry. Mercury emissions can be significantly reduced by implementing cleaner and more efficient industrial processes. This entails employing cutting-edge technologies and techniques to reduce or eliminate the use of mercury or mercury-containing substances in manufacturing processes.
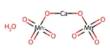
Sulphuric acid is the most commonly used chemical on the planet. It is an important reagent used in many industries, including paper, pharmaceuticals, and cosmetics, as well as batteries, detergents, and fertilizers. As a result, the fact that sulphuric acid frequently contains one of the most toxic substances – mercury – poses a global challenge. Researchers at Chalmers University of Technology in Sweden have developed a method that can reduce mercury levels in sulphuric acid by more than 90% – even from very low levels.
“There has never been a viable method for purifying finished sulphuric acid.” We are well below the current limit values with such a drastic reduction in mercury content. Such pure, high-quality sulphuric acid is in high demand in industrial applications, and it is an important step towards lowering environmental impact,” says research leader Björn Wickman, Associate Professor at Chalmers’ Department of Physics.
Sulphuric acid is produced either from petroleum sulphur or as a byproduct in mining industry smelters. In the latter case, naturally occurring mercury in the ore may end up in the finished products. Mercury can also be found in recycled streams from smelters.
Purifying the sulphuric acid as well prevents additional mercury emissions while allowing industry to operate more cost-effectively and produce a high-purity, non-toxic product. The next step will be to scale up the method into a pilot process that is closer to real-world volumes of thousands of tones.
Vera Roth
Toxic emissions that affect all life on Earth
Mercury dispersal is a worldwide problem, as the substance is volatile and can be dispersed by air over large areas. This toxic heavy metal is then washed into streams and lakes when it rains. It is stored in the soil, water, and living organisms, impacting the entire food chain. It can damage the brains and central nervous systems of humans and animals.
According to a UNEP report, mercury emissions into the atmosphere increased by 20% between 2010 and 2015. In 2015, approximately 2,200 tonnes of mercury were emitted into the atmosphere as a result of human activities such as cement manufacturing, small-scale gold mining, coal combustion, metal production, and other manufacturing industries. Furthermore, an estimated 1,800 tonnes of mercury ended up in the soil and water that same year. According to the report, mercury concentrations in the atmosphere may have increased by 450 percent over the last century.
“Any and all ways we can reduce mercury emissions are good, because any mercury that is emitted accumulates in the environment and continues to pose a health threat for thousands of years,” says Wickman.

Captures the metal using electrochemistry
His Chalmers research team presented a ground-breaking method for removing mercury from water using electrochemical processes five years ago. The method works by a metal electrode absorbing the toxic metal and forming an alloy. After that, the mercury can be safely removed and the electrode reused. The researchers have now taken this technology a step further, demonstrating how mercury can be removed from concentrated sulphuric acid in a new study.
The sulphuric acid experiments were carried out in collaboration with the mining and metals refining company Boliden and the company Atium, a spin-off from the Chalmers School of Entrepreneurship with the goal of bringing mercury removal from water and chemicals to market. The researchers now hope to be able to move forward with their partners and develop a type of reactor through which sulphuric acid can flow and be purified at the same time.
Potential to reduce costs and environmental impact
Today, mercury is mostly removed at an earlier stage – from the concentrates and recycled streams at the smelter before sulphuric acid is produced. This is an established process but leaves trace amounts of mercury in final products.
“Purifying the sulphuric acid as well prevents additional mercury emissions while allowing industry to operate more cost-effectively and produce a high-purity, non-toxic product. The next step will be to scale up the method into a pilot process that is closer to real-world volumes of thousands of tonnes,” says Vera Roth, a doctoral student at Chalmers and first author of the recently published article in the journal ACS ES&T Engineering.
Hoping for lower limit values
According to Statista, the global market volume for sulphuric acid is around 260 million tonnes per year. This figure is expected to rise to 314 million tonnes by 2029. The lower the sulphuric acid’s mercury content, the more valuable it is. When the mercury content of sulphuric acid for commercial purposes is less than 0.30 milligrams per kilogram, it is considered acceptable quality. Sulphuric acid is considered high purity if the content is less than 0.08 milligrams per kilogram. In their pilot study, the researchers used the new method to reduce the level of mercury to 0.02 milligrams per kilogram of sulphuric acid.
“The limit values for how much mercury sulphuric acid may contain are based on the technology available today. With the new method for purifying sulphuric acid, our hope is that the legislation around the limit values will be tightened in a global perspective where mercury levels are generally much higher,” says Wickman.
More information about the method and the study
The method of purification removes mercury from sulphuric acid by ionising the mercury and its ions, which form an alloy with another metal. Electrochemically, a platinum-surfaced electrode binds the mercury to itself. It then absorbs the toxic mercury and combines the two metals to form an alloy. The mercury can then be removed and the electrode regenerated in a controlled manner. This means the electrode can be reused and the toxic substance can be disposed of safely. The procedure is also extremely energy-efficient. Björn Wickman demonstrates how the method for removing mercury from water works in an episode of the Swedish Educational Broadcasting Company’s (UR) programme UR Samtiden.
Sulphuric acid is the world’s most used chemical. It is an important reagent used in many industries and it is used in the manufacture of everything from chemicals, paper, pharmaceuticals and cosmetics to batteries, detergents and fertilisers. It is an ingredient also for many recycling processes contributing into circular economy and extractive metallurgy.