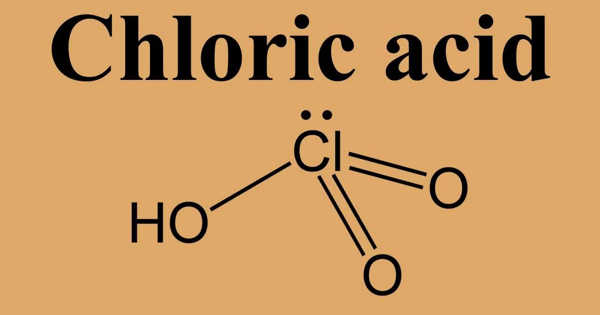
Chloric Acid, HClO3, is an oxoacid of chlorine, and the formal precursor of chlorate salts. It is an inorganic acid used as a reagent and reactive to prepare some chlorate salts and other chemical products. It is a strong acid (pKa ≈ −2.7) an oxidizing agent. It is a colorless liquid. It will accelerate the burning of combustible materials and can ignite most on contact.
Properties
Chloric acid is thermodynamically unstable with respect to disproportionation. It is a colorless liquid. Its density is 1 g mL-1. It is soluble in water. It is an oxidant and corrosive.
Chloric acid is stable in cold aqueous solution up to a concentration of approximately 30%, and a solution of up to 40% can be prepared by careful evaporation under reduced pressure. Above these concentrations, chloric acid solutions decompose to give a variety of products, for example:
8 HClO3 → 4 HClO4 + 2 H2O + 2 Cl2 + 3 O2
3 HClO3 → HClO4 + H2O + 2 ClO2
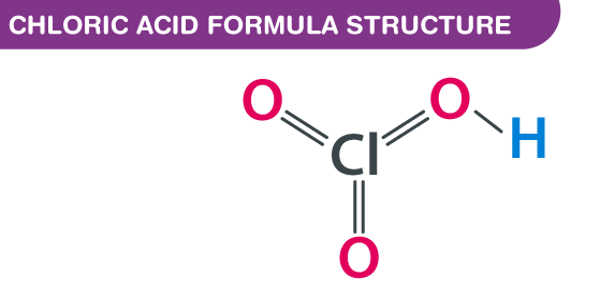
Formula and structure
The chloric acid chemical formula is HClO3 and the molar mass is 84.46 g mol-1. This salt is formed by one proton (H+) and one chloric anion (ClO–3). The chloric anion has a pyramidal-like structure, with a chlorine center atom to which two oxygen atoms are bound through double bonds and 1 oxygen atom is bound in a simple bond. The chlorine atom also has an electron pair that repealed the oxygen atoms and form the pyramidal structure.
Production
It can be prepared by the reaction of sulfuric acid with barium chlorate, the insoluble barium sulfate being removed by precipitation:
Ba(ClO3)2 + H2SO4 → 2 HClO3 + BaSO4
Another method is the heating of hypochlorous acid, producing chloric acid and hydrogen chloride:
3 HClO → HClO3 + 2 HCl
Uses
Chloric acid is used to prepare chloride salts and other chlorine species. It is used as a reagent in chemical analysis and to make other chemicals. It is also an oxidizer agent.
Hazards
Chloric acid is a powerful oxidizing agent. Due to its oxidizer characteristic, it should be used very carefully and must avoid contact with other chemical compounds that can react easily. Most organics and flammables will deflagrate on contact. Chloric acid is a strong acid, thus it can cause severe eyes and skin damage.
Information Source: