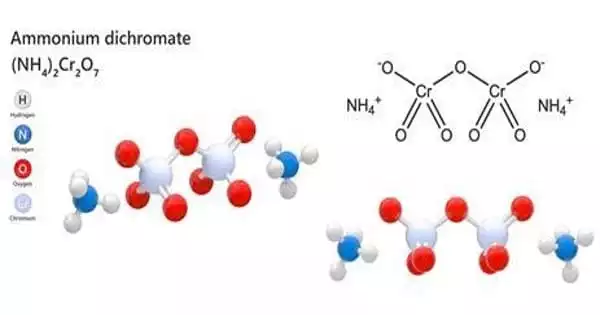
Ammonium dichromate has the formula (NH4)2Cr2O7 and is an inorganic chemical. It is made up of orange-red needles that form during crystallization. Chromium is in a +6 oxidation state in this compound, as it is in other chromates and dichromates, and is frequently referred to as hexavalent chromium. It is a salt made up of ammonium and dichromate ions. A hazardous chemical salt that is used to sensitize organic emulsions like gelatin to the hardening impact of actinic light. It is highly light-sensitive.
Because it is used in tabletop “volcano” presentations, ammonium dichromate is also known as Vesuvian Fire. However, due to the carcinogenic nature of the substance, this display has grown unpopular in schools. It was also employed in pyrotechnics and early photography.
Properties
At room temperature and pressure, the compound exists as orange, acidic crystals soluble in water and alcohol. It is formed by the action of chromic acid on ammonium hydroxide with subsequent crystallisation.
- Density: 2.12 g/cm³
- Molecular Weight/ Molar Mass: 252.07 g/mol
- Autoignition temperature: 190 °C
- Melting point: 180 °C
- Odour: Odourless
- Appearance: Orange-red crystals
- Stability: Stable under recommended storage conditions
- Specific Gravity: 2.1500
- Solubility: Insoluble in acetone; soluble in alcohol
The (NH4)2Cr2O7 crystal (C2/c, z=4) contains a single type of ammonium ion, at sites of symmetry C1(2,3). Each NH4+ centre is surrounded irregularly by eight oxygen atoms at N—O distances ranging from ca. 2.83 to ca. 3.17 Å, typical of hydrogen bonds.

Upon heating
Ammonium dichromate decomposes upon heating to produce nitrogen dioxide, water vapour (water is gaseous in this reaction due to the comparatively high reaction temperature), and solid chromium (III) oxide. It easily ignites and burns, leaving a thick green residue. If heated in a closed container, the container may rupture owing to material degradation. When coupled with or contaminated with combustible material, it can also operate as a potent oxidizing agent.
Uses
It has been employed in pyrotechnics, early photography, lithography, as a source of pure nitrogen in the laboratory, and as a catalyst. It is also employed as a mordant in the dyeing of pigments, the production of alizarin, chrome alum, leather tanning, and the purifying of oil.
- It is used in sensitizing solutions used in lithography.
- Used in pyrotechnics, lithography and photoengraving. It is also used as a magnetic recording material.
- It is used as an approved pesticide and used as a mordant for dyeing, used as a pigment.
Ammonium dichromate has been utilized in the laboratory as a source of pure nitrogen as well as a catalyst in pyrotechnics. This compound was also used extensively in the early days of photography and lithography. It is also employed as a mordant for dyeing pigments in the production of alizarin, chromium alum, leather tanning, and oil purification.
Safety
The Ammonium dichromate is similar to all the chromium (VI) compounds and it is highly toxic and also a proven carcinogen. Moreover, it is also a strong irritant at the same time.