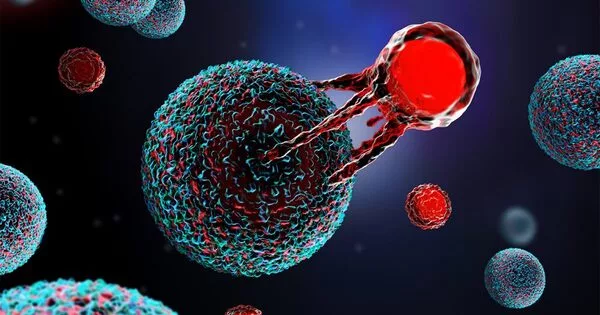
In the realm of cancer immunotherapy, the concept of developing a “suit of armor” for cancer-fighting cells such as T cells is an attractive one. While it may not be a real coat of armor, researchers have been working on several techniques to improve the body’s immune system’s effectiveness in targeting and destroying cancer cells.
Chimeric Antigen Receptor T-cell (CAR T) therapy has shown encouraging results in the fight against several types of cancer, but for many patients, the therapy causes severe and potentially fatal adverse effects. A research team has now discovered a technique that could allow CAR T therapies to realize their full potential while limiting severe side effects.
Cancer researchers have celebrated the emergence of chimeric antigen receptor T cell (CAR T) therapy in recent years, which has yielded encouraging results, altering the fight against numerous types of cancer. Patients’ T-cells are modified to target cancer cells, leading in extraordinary success rates for previously intractable kinds of cancer.
Six CAR T cell treatments have received FDA clearance, with several more on the way. However, these treatments have serious and potentially fatal side effects, including cytokine release syndrome (CRS) and neurotoxicity. These disadvantages emerge as a variety of symptoms ranging from high fever and vomiting to multiple organ failure and patient death, posing considerable barriers to greater clinical implementation.
Controlling CAR T-macrophage interactions in vivo is difficult. So, our study introduces a materials engineering-based strategy that involves incorporating a sugar molecule onto the surface of CAR T cells.
Michael Mitchell
Now, a research team lead by Michael Mitchell, associate professor in the University of Pennsylvania’s School of Engineering and Applied Science, has discovered a solution that could allow CAR T therapies to reach their full potential while limiting severe side effects. Their findings were published in Nature Materials.
“Addressing CRS and neurotoxicity without compromising the therapeutic effectiveness of CAR T cells has been a complex challenge,” Mitchell states. He claims that unfavorable interactions between CAR T and immune cells known as macrophages cause macrophage overactivation, which results in the release of toxic cytokines that cause CRS and neurotoxicity.
“Controlling CAR T-macrophage interactions in vivo is difficult,” Mitchell says. “So, our study introduces a materials engineering-based strategy that involves incorporating a sugar molecule onto the surface of CAR T cells. These sugars are then used as a reactive handle to create a biomaterial coating around these cells directly in the body, which acts as a ‘suit of armor,’ preventing dangerous interactions with macrophages.”
Ningqiang Gong, a postdoctoral researcher in the Mitchell Lab, explains the technique: “We attached this sugar molecule to the CAR T cells using metabolic labeling.” This alteration allows CAR T cells to assault cancer cells unhindered.”
“When symptoms of CRS begin to manifest, we introduce another molecule — polyethylene glycol (PEG) — to create the suit of armor, which effectively blocks dangerous interactions between these engineered T cells, macrophages, and the tumor cells themselves,” Gong continues.
Over time, small tumor antigens can still reach what the researchers call ‘PEGylated CAR T cells,’ slowly activating and expanding them without triggering the severe side effects associated with rapid activation and expansion. As the CAR T cells slowly expand, the surface density of PEG becomes diluted, progressively restoring their ability to interact with other cells.
The team says that their approach offers more than just a safety net for patients; it also opens up a new “therapeutic window” for treatment. This is made possible, Gong says, due to the size differences among tumor cells, CAR T cells, and macrophages. He says tumor cells and CAR T cells are typically smaller (ranging from 5-10 ?m) compared to macrophages (>20 ?m), and as the surface density of PEG on CAR T cells begins to dilute, interactions between CAR T cells and tumor cells are restored before interactions with macrophages.
According to Mitchell, this repair allows CAR T cells to target and kill cancer cells without generating macrophage overactivation, reducing the possibility of hazardous CRS symptoms and neurotoxic consequences.”We were able to modulate the interactions between CAR T cells and macrophages by incorporating the PEG buffer. This allows for a safer and more effective therapy,” Mitchell explains.
Aside from that, the team is looking into the possibility of using in situ PEGylation in various forms of cellular immunotherapies and potentially broader applications. “The implications could be far-reaching,” Mitchell claims. “We’re looking at a potentially universal approach to making cellular therapies safer for all patients.”