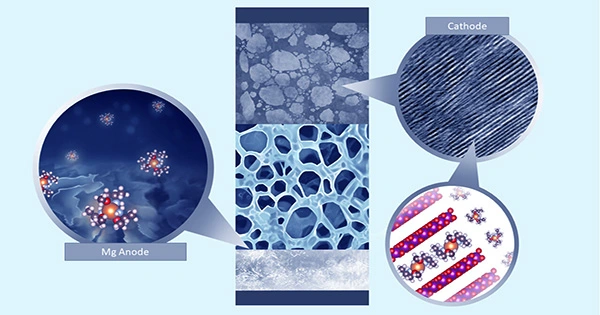
Engineers and material scientists are always working to increase the performance of battery technologies in order to meet the expanding needs of the electronics industry. Lithium (Li) metal batteries, or batteries having a metallic Li anode, are among the best-known and most extensively researched battery options.
While these batteries have been available for decades, researchers have recently attempted to increase their performance through the use of nanoengineering techniques as well as alternate solid or liquid electrolytes. The development of new potential electrolytes could considerably improve the stability and lifespan of Li metal battery cells.
Stanford University researchers recently published a new technique for developing high-entropy electrolytes for Li metal batteries. This method, described in a research published in Nature Energy, enabled them to find promising electrolytes that dramatically increase battery cycling stability at high current densities.

“Recent advances in electrolytes have greatly improved cyclability by enhancing electrochemical stability at the electrode interfaces, but achieving high ionic conductivity concurrently has remained challenging,” Sang Cheol Kim, Jingyang Wang, and their colleagues noted in their research. “We present an electrolyte design strategy for enhanced lithium metal batteries that involves increasing the molecular diversity in electrolytes, resulting in high-entropy electrolytes.”
The main goal of Kim, Wang, and their colleagues’ latest study was to make it easier to develop electrolytes with increased ion transport capabilities that do not jeopardize the stability of Li metal batteries. To do this, they developed a mechanism for modulating the architectures of solvents in weakly solvating electrolytes with substantial ion clustering.
As part of their research, they used X-ray scattering techniques as well as molecular dynamics (MD) simulations (computer-based simulations of molecular systems). This enabled them to uncover properties that make electrolytes for Li metal batteries particularly advantageous in terms of stability and ionic conductivity.
“We find that, in weakly solvating electrolytes, the entropy effect reduces ion clustering while preserving the characteristic anion-rich solvation structures, which are characterized by synchrotron-based X-ray scattering and molecular dynamics simulations,” Kim, Wang, and their colleagues wrote in their paper.
“When compared to conventional weakly solvating electrolytes, electrolytes with smaller-sized clusters exhibit a twofold improvement in ionic conductivity, allowing stable cycling at high current densities up to 2C (6.2 mA cm2) in anode-free LiNi0.6Mn0.2Co0.2 (NMC622)||Cu pouch cells.” Performance gains in three different weakly solvating electrolyte systems demonstrate the usefulness of the design concept.”
Kim, Wang, and their colleagues’ novel approach could soon aid in the development of a wide range of promising electrolytes for Li metal batteries, contributing to the ongoing search to improve their performance. These improved batteries power a variety of devices, including electric or hybrid automobiles, medical equipment, and other cutting-edge technologies.
“We anticipate that this work will stimulate efforts to develop HEEs that will bring lithium metal batteries closer to practical applications, as well as to broadly develop high-entropy solutions with superior properties for a variety of applications,” the researchers stated.