Antimicrobial resistance occurs when germs such as bacteria and fungi develop the ability to resist drugs that are designed to kill them. Treatment of resistant infections can be difficult, if not impossible. Antimicrobial resistance is a natural phenomenon. Antimicrobial resistance, on the other hand, is driven by a combination of germs exposed to antibiotics and antifungals, as well as the spread of those germs and their resistance mechanisms.
Antibiotics are medications that are used to kill bacteria. Certain groups of these germs may adapt to these medications over time. Antibiotics may no longer be effective against them. This is referred to as antibiotic resistance. Bacteria are microscopic organisms. They have the ability to enter your body. Some of them are harmless and may even be beneficial. However, some of these germs are dangerous. These can cause disease if they multiply inside your body.
It takes a long time to determine the drug susceptibility profile of a bacterial infection. Now, Nara Institute of Science and Technology researchers and collaborators have published reports on a technology that will dramatically accelerate this otherwise slow process and potentially save lives.
Although there was a misidentification error of less than 10% in our work, there was a clear discrimination between susceptible and resistant cells within 2 hours of antibiotic treatment.
Yoichiroh Hosokawa,
The CDC states that antibiotic-resistant infections are responsible for killing over a million people worldwide every year. Central to managing resistant infections is quickly identifying an appropriate treatment to which the infective bacteria are susceptible. “Oftentimes susceptibility results are needed much faster than conventional tests can deliver them,” says Yaxiaer Yalikun, senior author. “To address this, we developed a technology that can meet this need.”
The group’s work is based on impedance cytometry which measures the dielectric properties of individual cells with high throughput—more than a thousand cells per minute. Because the electrical readout of a bacterium corresponds to its physical response to an antibiotic, one has a straightforward means of determining whether the antibiotic works against the bacteria.
Conventional impedance cytometry involves analyzing the test (antibiotic-treated) and reference (untreated) particles in one sample followed by calibrating the impedance of the two particles—both steps require technical specialists to carry out extensive post-processing, which was a major limitation the group was determined to overcome.
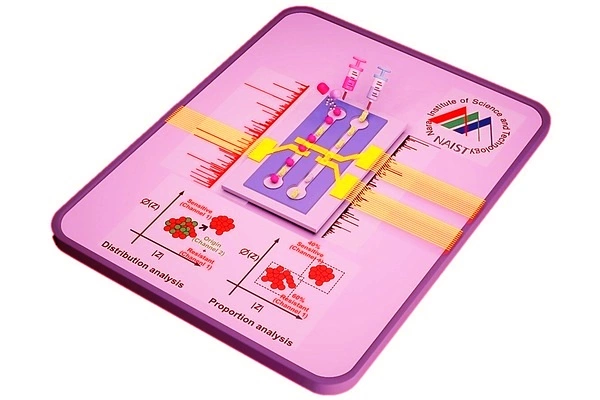
In a study published in ACS Sensors, the group develops a novel impedance cytometry method that simultaneously analyzes the test and reference particles in separate channels, creating easily analyzable separate datasets. This cytometry had nanoscale sensitivity, allowing for the detection of even minute physical changes in bacterial cells.
In a concurrent study published in Sensors and Actuators B: Chemical, the group designed a machine-learning tool to analyze the impedance cytometry data. Because the new cytometry method splits the test and reference datasets, the machine learning tool could automatically label the reference dataset as the “learning” dataset and use it to learn the characteristics of an untreated bacterium. By real-time comparison with antibiotic-treated cells, the tool can identify whether the bacteria are susceptible to the drug and can even identify what proportion of bacterial cells are resistant in a mixed-resistance population.
“Although there was a misidentification error of less than 10% in our work, there was a clear discrimination between susceptible and resistant cells within 2 hours of antibiotic treatment,” explains Yoichiroh Hosokawa, another senior author in the group.
This work is not limited to rapid evaluations of infections in clinical settings. For example, drug discovery researchers could use it to conduct quick initial investigations of drug efficacy against any cell, as long as the cellular response results in a change in dielectric properties. Impedance cytometry might become a staple of clinical and research labs in the coming years.