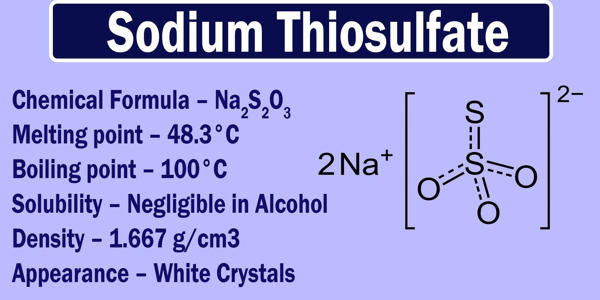
Sodium thiosulfate is an inorganic compound with the formula Na2S2O3.xH2O. It is typically found in its pentahydrate form which is either white in color, or colorless altogether. Typically it is available as the white or colorless pentahydrate, Na2S2O3·5H2O. In its solid form, it is a crystalline solid which has a tendency to readily lose water. The solid is an efflorescent (loses water readily) crystalline substance that dissolves well in water.
Sodium thiosulfate is used in gold mining, water treatment, analytical chemistry, the development of silver-based photographic film and prints, and medicine. It has a role as an antidote to cyanide poisoning, a nephroprotective agent, and an antifungal drug. The medical uses of sodium thiosulfate include treatment of cyanide poisoning and pityriasis.
Sodium thiosulfate is an inorganic sodium salt composed of a 2:1 mixture of sodium and thiosulfate ions. It has a role as a cyanide poisoning antidote, a nephroprotective agent, and an antimicrobial product.
Structure
Two polymorphs are known as the pentahydrate. The anhydrous salt exists in several polymorphs. Sodium thiosulfate is readily soluble in water and is also referred to as sodium hyposulfite. In the solid state, the thiosulfate anion is tetrahedral in shape and is notionally derived by replacing one of the oxygen atoms with a sulfur atom in a sulfate anion. The distance between the two sulphur atoms in the thiosulfate ion is comparable to the distance between two sigma bonded sulphur atoms. The S-S distance indicates a single bond, implying that the terminal sulfur holds a significant negative charge and the S-O interactions have more double-bond character.
Production
On an industrial scale, sodium thiosulfate is produced chiefly from liquid waste products of sodium sulfide or sulfur dye manufacture.
In the laboratory, this salt can be prepared by heating an aqueous solution of sodium sulfite with sulfur or by boiling aqueous sodium hydroxide and sulfur according to this equation:
6 NaOH + 4 S → 2 Na2S + Na2S2O3 + 3 H2O
Despite being stable at standard conditions, the sodium thiosulfate salt decomposes at high temperatures to yield sodium sulfate along with sodium polysulfide.
Uses
Sodium thiosulfate is used to treat cyanide poisoning. It is also used to reduce the side effects of the cancer medication cisplatin. It is on the World Health Organization’s List of Essential Medicines, the safest and most effective medicines needed in a health system. It is a very important chemical compound in the medical treatment of cyanide poisoning cases.
Information Source: