In a new study, researchers look into tangles in the brain, which are pathologies that are common not only in Alzheimer’s disease but also in other neurodegenerative diseases. The study focuses on Rbbp7, a protein whose dysregulation appears to be linked to the eventual formation of tau protein tangles and the rampant cell death seen in Alzheimer’s and other neurodegenerative diseases. Since the discovery of Alzheimer’s disease more than a century ago, two features of the devastating illness have taken center stage.
The first are amyloid plaques, which are dense accumulations of misfolded amyloid protein found in the spaces between nerve cells. The majority of efforts to slow the progression of Alzheimer’s disease have focused on amyloid protein plaques. To date, all have met with disheartening failure.
Until recently, the second classic trait received less attention. It is made up of string-like formations within the bodies of neurons that are produced by another important protein called tau. These are referred to as neurofibrillary tangles.
In a new study, researchers investigate tangles in the brain – pathologies not only characteristic of Alzheimer’s but other neurodegenerative conditions as well.
In a new study, researchers from the ASU-Banner Neurodegenerative Disease Center at the Biodesign Institute and their colleagues investigate these tangles in the brain – pathologies that are not only common in Alzheimer’s disease but also in other neurodegenerative diseases.
The study focuses on Rbbp7, a protein whose dysregulation appears to be linked to the eventual formation of tau protein tangles and the rampant cell death seen in Alzheimer’s and other neurodegenerative diseases.
“We had a hunch that this protein was involved in Alzheimer’s disease, especially since we know that the protein was decreased in Alzheimer’s disease post-mortem brain tissue when compared to normal brains,” says Nikhil Dave, the study’s lead author.
The study found a link between lower Rbbp7 levels and increased tangle formation, as well as associated neuronal loss and brain weight reduction in Alzheimer’s disease brains. Surprisingly, cell loss and tangle formation were reversed in transgenic mice with Rbbp7 levels restored to baseline.
The findings open up a new line of inquiry that could aid in the development of effective treatments for Alzheimer’s disease as well as a variety of tau-related diseases known as tauopathies, which include Pick’s disease, frontotemporal dementia, and traumatic brain injury.
The new study appears in the current issue of the journal Acta Neuropathologica.
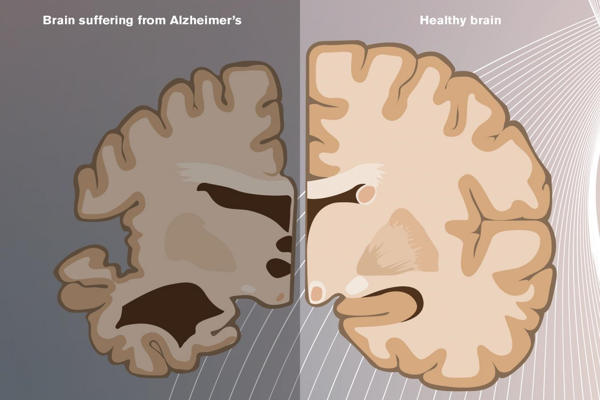
Infernal assault
Alzheimer’s disease is still one of the most puzzling diseases known to medical science. Its clinical symptoms appear gradually over time and can be masked by normal aging processes. However, once it has taken control of the brain, the disease’s progression is often swift and merciless.
Confusion, physical disorientation, delusions, forgetfulness, aggression, agitation, and progressive loss of motor control are some of the symptoms that patients may experience.
Alzheimer’s disease has been silently ravaging the brain for decades, according to researchers, by the time the first outward manifestations of the disease become apparent, typically leaving its calling card in the form of plaques and tangles.
Alzheimer’s disease is still the leading cause of dementia, with age being the primary risk factor. As life expectancy rises and other once-fatal illnesses become treatable, if not curable, the disease has been on a frightening upward trajectory. According to the Centers for Disease Control and Prevention, 5.8 million people in the United States alone suffer from Alzheimer’s disease, with the number expected to rise to 14 million by 2060.
Aside from advancing age, many other factors contribute to this complex disorder, ranging from hereditary predisposition to vascular diseases such as diabetes and obesity. Lifestyle choices, such as diet and exercise, can also have an impact on vulnerability. The disease typically affects people over the age of 65, though early-onset forms of the disease can strike much sooner.
Genome 2.0
The new study looks at yet another risk factor for neurodegenerative disease, this time focusing on an individual’s genes and how they are expressed. Although the three billion letter DNA code that makes up an individual’s genome remains fixed throughout life, scientists have discovered that chemical messengers of varying complexity and variety can act on the genome, delivering instructions to the DNA and guiding its behavior.
These epigenetic changes, as they are known, have the ability to turn genes on and off as well as regulate the amount of protein produced by these genes. Earlier ideas in biology that emphasized a static view of genomic destinies have given way to a new picture of life in which environmental changes can have a significant impact on how our genes behave. Scientists are only now beginning to understand the epigenome’s far-reaching impact on human health and disease.
The current study describes epigenetic changes that occur in the brain when the protein Rbbp7 levels are reduced, which the researchers discovered in post-mortem brain tissue from Alzheimer’s patients.
Rbbp7 regulates gene expression as one of its functions. It accomplishes this by altering the interaction of DNA with histone proteins, which DNA wraps around like sewing thread around a spool. When the DNA thread is loosely wrapped around the histone spool, the exposed DNA message can be read by the cell machinery and transcribed into mRNA, which is then translated into protein. However, if the DNA thread is tightly wrapped around the histone, the DNA genes are hidden from view, and transcription may be partially or completely blocked, reducing or disabling protein expression.
The researchers discovered that when Rbbp7 levels are reduced, the level of another protein known as p300 rises, resulting in acetylation of tau protein, a post-translational modification. This causes tau protein to detach from cell structures known as microtubules, which tau normally binds to. The freed tau is then free to accumulate within neurons, eventually forming the recognizable tangles associated with Alzheimer’s disease. (See the accompanying diagram.)
The acetylation of tau caused by low Rbbp7 leads to increased phosphorylation of tau, which promotes tangle formation and neuronal loss in the brain.
In the new study, transgenic mice with tau pathology had lower levels of Rbbp7 and more neuronal loss. Rbbp7 restoration to normal levels in mice reversed these pathologies, but cognitive deficits remained.
The reason for this, according to Ramon Velazquez, corresponding senior author of the new study, is that the study only targeted a small subregion of the hippocampus, while other brain areas associated with cognition were still rife with tangle formation. “In the future, we plan to look at the global effect of overexpressing Rbbp7 to see if we can rescue learning, memory, and other aspects of cognition.”
Light at the end of the tunnel?
The study’s findings regarding Rbbp7 levels and the formation of tau tangles, cell death, and cognitive function loss in the brain are compelling. The findings suggest that Rbbp7 could be a promising target for drug development and the development of effective therapies for Alzheimer’s disease and other tau-related diseases. Treatments based on such research may be ready for clinical trials within the next five years.
However, the authors emphasize that other molecular players are most likely involved in these complex processes. In the future, the researchers intend to conduct extensive, unbiased investigations into protein interactions, transcription pathways from DNA to mRNA, and epigenetic modifications that can lead to neurodegenerative disease.