The major mechanism underlying the transition from chronic to acute P. aeruginosa infections has been identified by researchers. Their findings can help to develop future treatments for life-threatening acute infections.
People with compromised immune systems are constantly at risk of infection. Pseudomonas aeruginosa, a common environmental bacterium, can colonize various body parts, including the lungs, resulting in chronic infections that can last a lifetime, which is common in people with cystic fibrosis. However, bacteria can occasionally change their behavior and enter the bloodstream, turning chronic localized infections into acute and potentially fatal infections. Despite decades of research in lab settings, how and why the switch occurs in humans remains unknown.
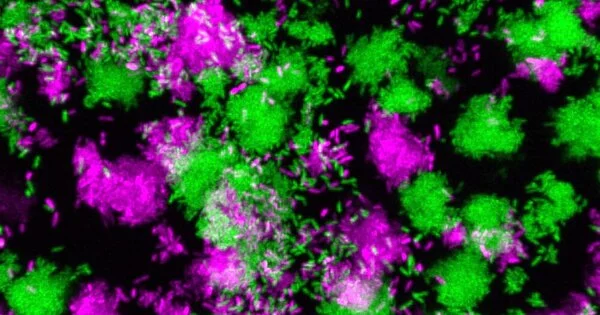
The major mechanism underlying the transition between chronic and acute P. aeruginosa infections, however, has been identified by researchers at the Georgia Institute of Technology. Marvin Whiteley, a professor in the School of Biological Sciences and the Bennie H. and Nelson D. Abell Chair in Molecular and Cellular Biology, and Pengbo Cao, a postdoctoral researcher in Whiteley’s lab, discovered the switch gene. The researchers discovered a biomarker for the transition by measuring bacterial gene expression in human tissue samples.
Their findings, which were published in Nature, can help to develop future treatments for life-threatening acute infections.
For years, people have been studying these bacteria in well-controlled lab environments, even though the lab is a place most microbes have never seen. Our study took a novel approach by looking directly into the bacterium’s behavior in the human host.
Marvin Whiteley
Bacteria, like animals, are adaptable and behave differently depending on their surroundings, according to Whiteley and Cao. A person with a chronic infection may be fine one day, but changes in the body’s environment can cause bacteria to change their behavior. This can result in an acute infection and sepsis, which requires immediate treatment.
“For years, people have been studying these bacteria in well-controlled lab environments, even though the lab is a place most microbes have never seen,” Whiteley explained. “Our study took a novel approach by looking directly into the bacterium’s behavior in the human host.”
The researchers chose to look at human tissue samples of chronic bacterial lung and wound infections. Using genetic sequencing technologies, Whiteley and Cao measured the levels of all types of mRNA present in the bacteria. The mRNAs encode the proteins that do all the work in a cell, so by measuring a bacterium’s mRNA level, one can infer the bacterium’s behavior.
While P. aeruginosa has approximately 6,000 genes, Whiteley and Cao discovered that one gene, known as PA1414, was more highly expressed in human tissue samples than all of the other thousands of genes combined. The levels were so high that Cao and Whiteley initially thought the amount of PA1414 mRNA was an artifact caused by the sequencing methods.
“This particular gene is not expressed very much in the standard lab environment, so seeing these levels was surprising,” Cao said. “At the time, the gene’s function was unknown.”
The researchers also discovered that low oxygen levels drive the gene’s high expression. As bacteria frequently experience oxygen deprivation during chronic infections, this is a common environmental feature of bacterial infections. Further research revealed that the gene also regulates bacterial respiration in low-oxygen environments. Interestingly, the researchers discovered that the gene encodes a small RNA that is essential for bacterial respiration rather than a protein. The small RNA was named SicX (sRNA inducer of chronic infection X).
The researchers then put the gene to the test in various animal infection models. They discovered that when SicX was absent, bacteria from chronic infections spread easily throughout the body, resulting in systemic infection. The researchers were able to conclude from the comparison that the gene is important for promoting chronic localized infection. Furthermore, researchers discovered that SicX expression decreased immediately during the transition from chronic to acute infection, implying that SicX may serve as a biomarker for the chronic-to-acute switch.
“In other words, without the small RNA, the bacteria become restless and go looking for oxygen, because they need to breathe like we need to breathe,” Whiteley said. “That need causes the bacteria to enter the bloodstream. Now, we know that oxygen levels are regulating this transition.”
A better predictor of when an infection might enter the bloodstream would represent a paradigm shift in treatment.
“If you can predict when an acute infection will occur, a patient could take a diagnostic test at home to determine if and when they may need to get treatment – before the infection becomes life-threatening,” Whiteley explained.
The research addresses long-standing questions about how and why chronic infections become acute. The findings of the researchers also open the door to the development of therapeutics that target this specific molecular behavior associated with P. aeruginosa infections.