International business activities at Biopharma Limited
Pharmaceutical industry is one of the most important manufacturing industries in Bangladesh. It has become one of the fastest growing sectors in Bangladesh. Presently the industry meets 97% of local demand and exports to more than 80 countries. The industry has been experiencing strong growth over the last few years. While the industry is achieving selfsufficiency, it yet procures 70% of raw materials from abroad. But developments are already taking place with a number of firms now manufacturing raw materials locally.
History of Pharmaceutical Industry:
The history of pharmaceutical industry dates back to 1950s. Over the years it has gone through few changes. After the post liberation war 1971 the industry was largely dominated by MNCs and the country was highly import oriented. In 1982, through the formulation of national drug policy and drug control ordination, a defined guideline for the development of the industry was created. By then 75% of the market was dominated by the MNCs, whereas the rest were shared by some 133 local firms. Since then, the, local firms have established a strong foothold and it has transformed to an active exporter from an import dependent of pharmaceutical industry. In 2010, top 5 MNCs have approximately 9.05% of the market share and 97% of the total local demand is met through local production.
Objectives:
Primary Objective:
My main objective is to understand the different etiquettes and activities regarding the international organizational operations in pharmaceutical companies in Bangladesh, significantly in Biopharma Ltd.
Secondary Objectives:
Along with the primary objective, the following things are also included with the report:
- To know how a pharmaceutical company does business development for international business
- To understand pharmaceutical company negotiation and contract signing
- How different financial and freight procedure are operated
- Ways of dealing buyer and objection handling
- To know about documentation system for international business
Scope of the Report
The scope of the report is to bring to light the actions BPL needs to ponder over in respect of its training and development system to attain the desired standard of product quality to penetrate into the high regulated market of the developed countries.
Quality in this context means a product which is pure and which is consistent. Medicine needs to avoid any kind’s contamination from other substance since this could have damaging effects upon the person taking medicine.
Data Analysis
- Primary Data- It was derived from the practical task of the work such as communication with the staff and discuss with the manager of international business department.
- Secondary Data- Internet, Office Record, Different Articles.
Finding of the Data
The collected data were analyzed and were pointed out and also shown as finding at the end.
Background of the Company:
Biopharma Limited is one of the leading manufacturers of pharmaceutical finished products of Bangladesh having WHO cGMP manufacturing facilities. It is committed to serving mankind with the products having strict standard of quality, safety and efficacy to ensure public healthcare through uncompromising scientific and professional approach complying with the regulatory requirements of WHO cGMP and Quality Management System. It is also an ISO 9001: 2008 Certified Company. By virtue of the highest quality of products, the company has already gained the trust and confidence in healthcare professionals and patients all the country. Biopharma is always committed to assure the best quality pharmaceutical products and best services to the customers. It started its journey in 1999 with the pharmaceutical finished products by the involvement of a group of inspired physicians having innovative mission and global conception. It is a Private Limited Company registered under Registrar of Joint Stock Companies. Its technical experts (pharmacist, Chemists, Biochemists, Microbiologists Analysts and other professionals ) skilled and trained staffs always try to leave no stone unturned in their professional works by following WHO cGMP guidelines and the guidelines & instructions of the Drug Administration & Licensing Authority of Bangladesh to ensure the production of quality medicine. Good Laboratory practices (GLP) have always been performing with a strict discipline to follow their professional ethics.
Future Prospect of BIOPHARMA Ltd
BIOPHARMA is striving to improve its existing facility by carrying out regular inspection and updating of technology for its products and processes. BIOPHARMA is emerging with its world class manufacturing facility complying with the regulatory requirements of WHO cGMP, US-FDA, UK-MHRA, EMEA and Australia- TGA. A comprehensive plan has also been taken by Bio Group to set up an Active Pharmaceutical Ingredients (API) Plant. Bio Group looks at the future with increasing confidence. The time to come Bio Group intends not only to strengthen its strong local footing but also to expand its presence in the international markets.
BIOPHARMA Ltd. has already starts its second production plant in Narayangong in addition of a cost of Tk. 194 crore. After completion of the project BIOPHARMA Ltd. will contribute about 39% of the market gap of demand of pharmaceutical product and also contribute to the country API market.
Top Pharmaceutical Companies in Domestic
Presently top pharmaceutical companies in Bangladesh are also in the process of getting into bulk drug production with collaborative technology, technology transfers and joint venture basis. The large-scale players in the Bangladesh pharmaceutical industry currently include Square Pharma, Incepta, Beximco, Opsonin, Acme, ACI, Aristopharma, SK+F, Reneta, Drug International, Orionpharma, Biopharma and among others. The MNCs that have a major presence in the country’s pharma sector are Aventis, Pfizer, Novartis, Glasko-Smith and Astra Zeneca.
Top Pharmaceutical Companies in Export
Pharmaceuticals company now getting huge drug production with use of high technology, Skillful employees and joint venture basis. After meeting local market most of the pharmaceuticals company now exports their product in abroad. The large-scale players in the Bangladesh at export pharmaceutical industry currently include Novarties (Bangladesh)Ltd, Beximco Pharmaceuticals ltd, Square Pharmaceuticals ltd, Incept Pharmaceuticals ltd, The Acme Laboratories Ltd, Eskayef(Bangladesh)Ltd , Renata Ltd, Jayson Pharmaceuticals ltd, Aristropharma ltd, Biopharma Limited, Opsonin Pharma ltd etc.
Exporting countries name
Most of the pharmaceuticals company now export their drugs to foreign country after fulfill the demand of local market. Biopharma is also export their drugs in foreign countries. And Biopharma has taken 10th position who exports pharmaceuticals products in different countries. Countries name are; Afghanistan, Bhutan, Sri Lanka, Myanmar, Vietnam, Philippines, Guam, Kenya, Nigeria, Mali & Liberia.
International Business Activities of Biopharma
Business Development:
Business development is a very important tool with which a company expands its business to other markets and creates a potential market for its own. Therefore, a company should be very careful about this matter. In order to develop a business, a company needs to know some basic information about the environmental forces, i.e. demographic, political, economical, socio-cultural, legal, and technical and so on.
Business Etiquettes and Communications:
It is very important to know proper business etiquettes and manners for the international business students. Due to working in Biopharma, I learnt those etiquettes more clearly and concisely. Communication manner is also an important practice for all business personnel. I learned written communication through which a pharmaceutical company creates business relationships and involves with corporate activities. The etiquettes of walking, talking, body posture, dining or presenting- all matters for any corporate person and I have learnt it there.
Agreement and Authorizations:
In order to operate business in abroad, first thing comes is a buyer or an importer who will sell the products in its country. For doing this there should be an agreement by which the parties would agree to do business together with trust. This agreement mentions all the rights and obligations, terms and conditions regarding partnership for both the parties. After that there comes the question of authority. To solve the problem, the buyer prepares a “Power of Attorney” where it is stated that the opposite party has been given rights to market the goods registered for trade.
Negotiation and Dealing with Clients:
Negotiation is a very common affair in any business activity. In Biopharma, I studied some negotiation incidents as well. From the study I learned that both companies always try to win the game through negotiation. Sometimes, negotiation reaches to a dead end and after that people come again and try to find out the best possible situation which would be profitable for both th. In order to develop a business, a company needs to know some basic information about the environmental forces, i.e. demographic, political, economical, socio-cultural, legal, and technical and so on.
In Biopharma, I studied some negotiation incidents as well. From the study I learned that both companies always try to win the game through negotiation. Sometimes, negotiation reaches to a dead end and after that people come again and try to find out the best possible situation which would be profitable for parties. In order to get better profit than the opposite party negotiation skill really matters. In this case, experience is really like an asset.
Shipping Procedure:
Though shipping procedure is a vast thing and it is not possible to understand the whole thing in this short time. However, I managed to observe these situations and did some tasks which reduced my fear about the complicated processes and enriched my ability and experience. For a shipment one needs to know some basic things without which it would be nearly impossible to operate a shipment. Those things are:
- Type of Payment:
Payment type can be of three types; L/C, T/T and document against payment. L/C stands for Letter of Credit; a definite of issuing bank on behalf of the importer to the exporter’s bank that a certain amount to be paid after complying documents. It is used for larger quantity order shipped by sea. T/T stands for Telegraphic Transfer. It is the most simple and easiest payment method to use. It is used for sample and small quantity shipments sent via air. On the other hand, documents against payments are used when exporter makes shipment and sends the shipping document to the importer’s bank for collection. The major advantage of the cash against documents payment is the low cost compare to the L/C.
- Incoterms:
Incoterms or International Commercial Terms are a series of international sales terms, published by the International Chamber of Commerce (ICC). There are 13 types of incoterms. It is very important to understand the types of payments and responsibilities between buyers and sellers and reflects the state-of-the-art transportation practices. These are – EXP (Export Works), FCA (Free Carrier), FAS (Free Alongside Ship), FOB (Fright on Board), CFR (Cost and Fright), CIF (Cost Insurance and Fright), CPT (Carriage Paid To), CIP (Carriage Insurance Paid to), DAP (Delivered at Place), DAT (Delivered at Terminal), DES (Delivered at Frontier), DEQ (Delivered Ex Quay), DDU (Delivered Duty Unpaid), DDP (Delivered Duty Paid).
Among all these incoterms, CIF (Cost Insurance and Fright), CRF (Cost and Fright) and FOB (Freight on Board) are used widely where EXW (Export Works) or DDP (Delivered Duty Paid) are not usually used.
- Bill of Lading and Airway Bill:
Bill of lading (BL) is a document issued by sea carrier of goods on receipt of cargo from the shipper. On the other hand, airway bill or AWB is issued by air carrier of goods after completion of export customs formalities. The major difference between them is AWB is not a document of title.
A bill of lading informs the names of consignee, shipper and notify party, place of receipt, name of port of loading, place of deliver, port of discharge, voyage no., container no., EXP no., BL/AWB/Contract no., description of the goods, manufacture and expire date, quantity, measurement, either shipment cost is prepaid or postpaid and so on.
- Paper Submissions:
Different departments need different documents, at the same time; number of copies also matters. It was quite a difficult and confusing thing if one does not understand; like how many copies of invoice or packing list do it need to submit to the bank, or to the buyer or to the customs or to the company itself for records. Same happened to me but when I started helping with those things I was able to understand those things quite well.
- Certificate of Origin (COO):
Certificate of Origin identifies goods and contains an export certificate by a government authority, or other empowered body, that the goods in question originate in a specific country. Many overseas importers insist upon a COO when dealing with Bangladeshi exporters. In Biopharma I also helped with preparing the COO and that was very exciting for me. For collection of a COO, one has to apply for a COO form with the commercial invoice for which the COIO is needed and tk.100 as fee from the Dhaka Chamber of Commerce and Industries (DCCI).
A COO includes some information like the names of both the parties, the of destination, transport medium, invoice no., EXP no., BL/AWB/contract no., commodity code, description of the goods, its weight and quantity and the value of the goods.
Banking and Customs Procedure:
In order to get bank clearance, I have learnt what papers do it needed to submit and the number of copies. Same was with the customs clearance. It might seem very easy but to me, at first, it was seemed very complicated when I was studying them. Later when I exercise those things practically, I was able to understand the whole process.
Documentation:
Documentation is very important tasks to be done. It is simple but very complex series of job. I did for sorting papers for buyers and office. I did sorting papers for pre-shipment as well.
For this I have learnt about Form 10 A, EXP, Insurance, Packing list, Commercial Invoice and others.
Organization wide:
At the international business department of Biopharma Ltd., very few things were left that I didn’t do. I have done almost every possible staffs which are equally important in every organization including pharmaceutical companies in dealing with foreign delegates. The international business department of Biopharma does many tasks. It starts its functions from a contract and ends up after the delivery the goods. In between, each and every task is operated and controlled by this department. In other sense, the international business department of Biopharma Ltd. does the exact jobs what a product marketing department does. I have experienced that the key difference is simply the market area of the departments.
The operation is not divided into any official sectors. The tasks are accommodated according to the efficiencies of the employees and it is enough for the organization. It does its job through some processes. These are described in a very short way:
- Continuous Communication:
The first step for an international trade is to create a communication in order to go further operations. Both parties communicate and then engage into a contract.
- Product Description:
After establishing a regular communication process, Biopharma provides necessary information and sends the product description so that the buyer can select its necessary items.
- Sample Sending:
After receiving the buyer’s choice; Biopharma sends the sample of those goods for further processes.
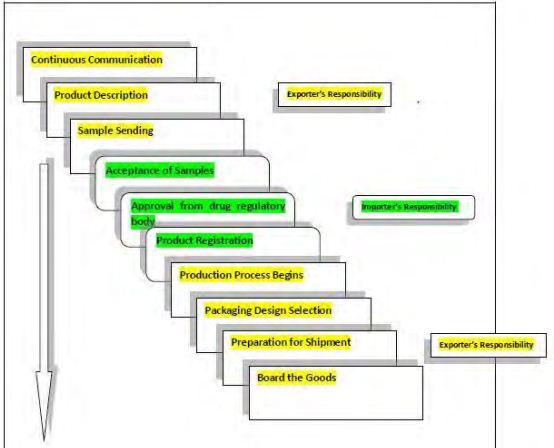
When the buyer finalizes the products that it wants to trade; it takes approval from the drug regulatory authority. If it does not have separate drug regulatory authority; it takes approval from the ministry of health.
4. Product Registration:
After the approval; the buyer goes for registration for the goods. Usually it takes two to three months to register the goods. When the registration process over; buyer informs it to Biopharma.
5. Start of the Production Process:
After the confirmation from the buyer; Biopharma starts the production procedure where it manages and updates all the functional activities and keep the buyer updated.
6. Packing Design Selection:
Packing design is a side-by-side task which is done on the basis of the interest of the buyer. The language, goods packing system everything are determined in here.
7. Preparation for Shipment:
Now all documents are collected and placed according to requirements. These things should be carefully handled because if anything goes wrong; it will affect the schedule of the shipment. So these things should be done so that company can avoid bad name.
8. Board Goods
This is the last step loading the goods for delivery to the buyer. At this stage company works with the post shipment documents.
Findings And Analysis
Observation at Biopharma Ltd.:
Every organization is composed of some internal strengths and weaknesses and also has some external opportunities and threats in its whole life cycle. It is same with Biopharma Ltd. The followings will briefly introduce the internal and external forces that I have explored in the past 3 months.
SWOT Analysis of Biopharma Ltd.:
Strength:
Efficient and Experience Management Team: Biopharma Ltd. is operated by a bunch of very efficient and effective management team. They work very hard to take the company to its high peak.
Devotion to Work: The devotion of doing the best has always been seen among the employees of this department. They have made it possible to maintain such important department.
Marketing Performance: The global marketing performance is very good which is one of the most important tools for successful international business operations.
Learning Curve: It is obvious; to be successful business organization, experience matters a lot and in international negotiation it is a must. Biopharma is very efficient in this case.
Highly Efficient Employees: IBD of Biopharma is blessed with a team of highly efficient employees who work hard to keep their commitment with their organization and indeed the importers.
Available Cash on Hand: Biopharma is well aware about the importance of good relationships between its buyers. That is why, international business department has sanctioned with availability of cash to deal with the foreign parties.
Weakness:
Inadequate services: There are many services offered from Biopharma which are not adequate in all places. It affects the company severely due to dissatisfaction from the customers.
Low HR strength: The HR of Biopharma is not up-to-the-mark. There are many things that should be repaired. Due to the poor HR performance, employee activity gets slower and causes huge damages.
Increased Raw Materials: Prices of raw materials are increasing day by day which increases the product costs both in home and abroad and may prove to be a weakness.
Opportunity:
Good Global Market Reputation: Biopharma is growing a good reputation regarding the international relationship. This happens due to the brotherly relationships between the two parties.
Increasing Client Reliability: Biopharma has goodwill of good client reliability. That is the reason; buyers are always interested in doing business with Biopharma.
Bright Future for Expansion: The reason pharmaceutical industry is rising is its low cost. That is why; importing countries are always interested in importing goods from Bangladesh. In this regard, Biopharma may grab the opportunity through its present network.
Market Expansion: Biopharma is expanding business through increasing market for business rapidly and it is a good sign for making a bright possibility for a successful organization.
New Therapeutic Area: Biopharma has a good potential regarding new therapeutic area to increase its product lines and spread its market.
Threat:
Very Competitive Market: Pharmaceutical industry has become a very potential market in Bangladesh. That is why; more and more companies are coming in and that create the market more competitive.
Political Instability Affects The Operations: Like other organization, Biopharma also affected by the political instability. Due to political instability, it becomes very tough to deliver goods to its customers.
Competitors have More Product and Services: The competitors of Biopharma have more products than it and it is a big disadvantage. This may prove a big factor for market failure.
Increased Bargaining Power of the Suppliers: Bargaining power of suppliers is increasing in the case of Biopharma Ltd.
Problems Regarding Working Place:
There are some general problems which should be addressed to make the working place more positive and qualitative. In this purpose; separate working place is required the most. But in Biopharma; employees from different departments working together and that create chaos and unsound for doing jobs. Therefore, working area should be separated. There are also few employees who spend the working hour by doing nothing which should be identified, and take proper steps.
Form 10 A:
Form 10 A is a document granted by the Directorate General of Drug Administration (DGDA) of Bangladesh which issues licenses for registering goods for export in abroad. It grants the quantity of the registered items and it has two years validity. In a Form 10 A, there are names and descriptions of goods, their packing size and quantity with which the calculation is done.
EXP Form:
Exp form is a document collected from the bank driven by the Bangladesh Bank. The main purpose of EXP form is to give judicial right to the business export. An EXP form. Contains the type of commodity, port and country of destination, quantity unit and volume, name of currency and amount of shipment, names and addresses of importer and exporter and some others.
Pro-forma Invoice:
A pro-forma invoice is a document that is sent to a buyer in certain circumstances- usually before all of the invoice details are known. It notes the kind and quantity of goods, their value and other important information regarding the goods. It is not a true invoice rather it is simply a document that declares the seller’s commitment to provide the goods or services specified to the buyer at certain price. It is also be used to declare the value of goods for customs.
Agreement Terms:
There are many terms in agreement which should be known to any business student. These are the preambles, definition of the terms, who appointments whom, registration, protection for intellectual property, terms and condition for indemnification, force majeure to be considered, preservation for confidentiality, terms and termination, arbitration policy, language to be used for communication, payment type, marketing and distribution policy, governing laws and other clauses.
SWIFT:
Society for Worldwide Interbank Financial Telecommunication or SWIFT provides a network that enables a financial institution worldwide to send and receive information on financial institutions. It is a member owned cooperative through which the financial world conducts its business operations with speed, certainty and confidence. More than 10,500 banking organizations, securities institutions and corporate customers in 215 countries trust it every day to exchange the millions of standardize financial massages.
RECOMMENDATION & CONCLUSION
Recommendation for Future Strategic Action:
I can suggest few recommendations for the future strategic action on the basis of working in Biopharma Ltd.
- More Employee Training Programs: More professionalism makes more efficient human resource. That is why; Biopharma should conduct more and more training program so that they will be more professional and efficient to deal with any problematic situations.
- Monthly Seminars: Biopharma should arrange monthly seminars or workshops as a medium of continuous interactions in order to building motivation among the employees. These seminars may make the owner and worker closer and create a strong bonding of trust.
- Knowledge over Foreign Culture: International business supposed to be working with international parties and different countries have different norms and believe. Therefore, it is very important to understand the importance of knowing their culture and traditions.
- Cross-cultural Communication: Training on cross-cultural communications should be provided for increasing professional skills.
- Flexible functional policies: More flexible functional policies are needed to match the changing environment of the industry.
- Emphasis on Foreign Language: Another important factor is the knowledge about foreign languages. For this purpose, learning English is a must. However, knowing French or Russian or Chinese would not be a waste of time. Language is not only for communication; it is a part of showing respect towards others cultures and togetherness. That is why language is given importance for making newer bonding.
- Patent Right: Patent right protection is mandatory to do business.
- Search for cost effectiveness: Looking for cost effective sources of raw materials to bring down the production cost.
Conclusion
The pharmaceutical industry in Bangladesh is gifted with unparalleled potential to grow in the days ahead as it enjoys a number of competitive advantages. After the end of the report, I have learnt many things about the international business dealings. The International Business Department of Biopharma Ltd. indeed a good example for international business operations for a pharmaceutical company. I have associated with many tasks regarding different shipments through making CO; documentations for the buyers, bank or office copies. Those were very nice and interesting tasks that I had experienced while working at Biopharma International Business Department.