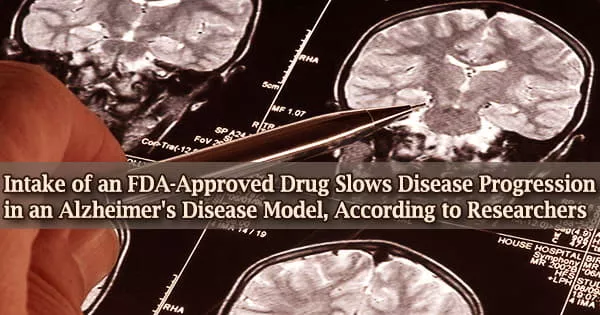
When employed in lab models, Indiana University School of Medicine researchers discovered that niacin slows the advancement of Alzheimer’s disease, a finding that could pave the way for medicinal approaches to the condition.
Niacin modifies microglia reactivity to amyloid plaques in an Alzheimer’s disease animal model, according to a new study published in Science Translational Medicine.
Gary Landreth, Ph.D., the Martin Professor of Alzheimer’s Research, and Miguel Moutinho, Ph.D., a postdoctoral scholar in Anatomy, Cell Biology, and Physiology directed the research.
“This study identifies a potential novel therapeutic target for Alzheimer’s disease, which can be modulated by FDA-approved drugs,” Moutinho said. “The translational potential of this strategy to clinical use is high.”
Alzheimer’s disease is a progressive brain disease that wreaks havoc on memory and thinking skills, as well as the capacity to carry out even the most basic tasks. Symptoms of late-onset type occur in the majority of patients with the disease in their mid-60s.
Early-onset Alzheimer’s disease is extremely rare and occurs between the ages of 30 and 60. The most prevalent cause of dementia in elderly people is Alzheimer’s disease. Alzheimer’s disease is expected to affect 60 percent to 70 percent of the approximately 50 million people globally who have dementia.
Niacin is a B vitamin that helps the body’s metabolism run smoothly. It can also be found in supplements and cholesterol-lowering medications. The brain, on the other hand, uses niacin in a different way, according to Moutinho.
This study identifies a potential novel therapeutic target for Alzheimer’s disease, which can be modulated by FDA-approved drugs. The translational potential of this strategy to clinical use is high.
Miguel Moutinho
Niacin interacts with HCAR2, a highly selective receptor found in immune cells physically linked with amyloid plaques, in the brain.
When the FDA-approved Niaspan medication, which was employed in this study, activates the receptor, it increases favorable immune cell responses, according to Landreth.
“After the Alzheimer’s disease animal models received niacin, they ended up with fewer plaques and they have improved cognition,” Landreth said, “and we directly showed that these actions were due to the HCAR2 receptor.”
If current population trends continue, the number of persons with Alzheimer’s disease will rise dramatically unless it can be efficiently treated or avoided. This is because the most important recognized risk factor for Alzheimer’s disease is getting older.
Forgetting recent events or discussions is one of the first indicators of Alzheimer’s disease. A person with Alzheimer’s disease will develop severe memory impairment and lose the capacity to carry out daily duties as the disease develops.
There is currently no cure for Alzheimer’s disease, while substantial progress has been made in researching and testing new therapies in recent years. The US Food and Drug Administration has approved several medications to treat Alzheimer’s disease.
Previous epidemiology studies of niacin and Alzheimer’s disease found that those who consumed more niacin in their diet had a lower risk of developing the condition, according to Landreth. Niacin is also being tested in Parkinson’s disease and glioblastoma clinical trials.
Landreth and Moutinho are partnering with Jared Brosch, MD, associate professor of clinical neurology, who is applying for a clinical pilot trial to explore the effects of niacin on the human brain to continue their research.