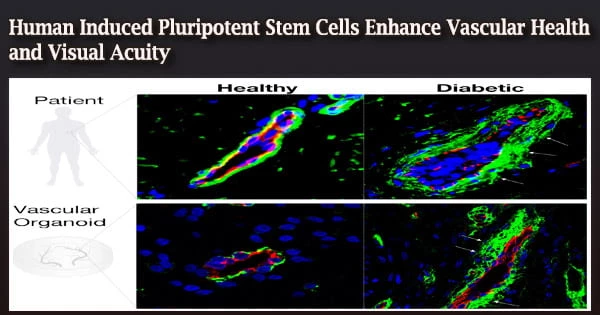
In order to better manage the vascular health issues caused by type 2 diabetes, researchers at Indiana University School of Medicine are working with the University of Alabama at Birmingham and five other institutions. Their work could one day support blood vessel repair in the eye among diabetic patients with early retinal vascular dysfunction.
In order to develop or mature human-induced pluripotent stem cells (hiPSCs) into the particular mesoderm subset of cells that exhibit vascular reparative capabilities, new techniques must be discovered and used.
“Vascular diseases afflict hundreds of millions of people in the world,” said Chang-Hyun Gil, MS, PhD, a postdoctoral fellow in the Department of Surgery and co-first author of the study. “In this study, we focused on the retinal vessel in type 2 diabetes. Our results demonstrate the safe, efficient, and robust derivation of the hiPSC-derived specific mesoderm subset for use as a novel therapy to rescue ischemic tissues and repair blood vessels in individuals with vascular diseases. The results provide a foundation for an early phase clinical trial.”
Researchers genetically reprogrammed diabetes and non-diabetic peripheral blood cells into hiPSCs and developed the cells into specialized blood vessel reparative cells in the multi-site, early phase work that was just published in Science Advances.
With the restoration of vascular perfusion, the effects of injection into animal models of type 2 diabetic murine (T2D) retinal dysfunction demonstrated a considerable improvement in visual acuity and electroretinograms. They postulated that endothelial progenitors with in vivo vessel reparative capabilities may be obtained from hiPSC-derived vascular reparative cells in these diabetic people.
“Unlike the use of embryonic stem cells (ESCs), genetically engineered hiPSCs do not carry the ethical challenges ESCs possess that limit their possible usage, and hiPSCs are being increasingly recognized as a viable alternative in study design and application as a cell therapy for human disorders,” Gil said.
As stated by the U.S. According to the Centers for Disease Control and Prevention, more than 37 million Americans, or more than 11% of the population, have diabetes. Furthermore, problems linked to diabetes have become more prevalent in both adults and young adults (aged 18 to 44 and 45 to 64, respectively).
In this study, we focused on the retinal vessel in type 2 diabetes. Our results demonstrate the safe, efficient, and robust derivation of the hiPSC-derived specific mesoderm subset for use as a novel therapy to rescue ischemic tissues and repair blood vessels in individuals with vascular diseases. The results provide a foundation for an early phase clinical trial.
Chang-Hyun Gil
Through disruption to the small and big microvessels that supply these tissues, these problems lead to significant metabolic changes that impair the cardiovascular, ocular, peripheral nerve, and renal systems.
More than 11% of persons 18 and older reported having significant vision problems or being blind in 2019, while more than 1.87 million adults had a major cardiovascular disease diagnosis.
“This work by Dr. Gil represents a monumental step forward in the application of induced pluripotent stem cells in treating the complications of diabetes,” said Michael P. Murphy, MD, the Cryptic Medical Research Foundation Professor of Vascular Biology Research at IU School of Medicine, a vascular surgeon at IU Health and Eskenazi Health and a co-author of the study.
Researchers isolated a specific mesoderm subgroup from hiPSCs to produce endothelial cells with vessel-repair capabilities that are comparable to those of endothelial colony-forming cells (ECFC).
Endothelial cells are cells that line the inside of blood arteries, lymph vessels, and the heart. They play a significant role in controlling inflammatory responses and vascular function. The movement of proteins from the blood into tissues is regulated by endothelial cells, which also control blood flow.
Gil claimed that mesoderm populations reverse vasodegeneration of damaged retinal capillaries and that the specific mesoderm subset expressing KDR, NCAM1, and APLNR (KNA+ mesoderm) demonstrates improved capacity to develop into ECFC and produce functional blood vessels in vivo. Studies using optokinetic nystagmus demonstrate increased vision, while electroretinograms suggest improved neuronal retinal function.
“The next translational step of the work is to transfer the research protocols reported for differentiation of the hiPSC into to S-KNA+ cells into large-scale manufacturing processes,” said Mervin C. Yoder, MD, distinguished professor emeritus and research advisor for the Indiana Center for Regenerative Medicine and Engineering.
Yoder is a second co-author of the paper and the scientific founder of Vascugen, a business devoted to developing therapies for illnesses that can be fatal and are brought on by microvascular issues.
“Selected aspects of this work have been licensed by Vascugen, Inc., through the Indiana University Innovation and Commercialization Office, who are focused on developing vascular reparative cells from induced pluripotent stem cells.”
Maria B. Grant, MD, a former Marilyn Glick Professor of Ophthalmology at the Eugene and Marilyn Glick Eye Institute and current endowed chair of ophthalmology at UAB, is a second study co-author.
As a follow-up to a grant she and Yoder have been working on for the past 20 years involving stem cells and how they might be used to repair blood vessels in the eye, Grant said this study is extremely translational.
Although it can take a while for hiPSCs to develop, the study team shortened the procedure to speed up the development and increase the likelihood that they could be used as a human therapy to repair blood vessels in the eye.
“At UAB, we took the stem cells and hiPSC cells and studied them,” she said. “Science is really team science. I bring all the eye experience and some stem cell experience, and Dr. Yoder brings a lot of stem cell experience. It’s a complementary collaboration.”
“I want to express my greatest thanks to Dr. Yoder, Dr. Grant, and Dr. Murphy for their support,” Gil said. He will remember Yoder for “his dedication, passion, patience and kindness” and Murphy for supporting him in completing the study.