Migraine pain signals are generated when the nerves in the brain become overexcited, causing inflammation and activation of pain receptors. The exact mechanism of how this happens is not fully understood, but it is thought to involve a combination of genetic and environmental factors.
Schwann cells, which are abundant in the peripheral nervous system and form a protective sheath around nerve fibers, have been discovered to play an important role in migraine pain by an international team of researchers. Their research, which was carried out in mice and human Schwann cells and published in Nature Communications, shows how pain is signaled from within Schwann cells and identifies several ways to block this signaling, providing potential targets for new migraine treatments.
Migraines affect more than 15% of adults, with women being twice as likely as men to suffer from these severe headaches. Calcitonin gene-related peptide (CGRP), a small protein in the nervous system, is known to play an important role in migraine pain; in fact, a new class of migraine drugs uses monoclonal antibodies to target CGRP or its receptor.
“While CGRP has been linked to migraine pain, the mechanism by which it causes pain has been a source of debate in the scientific community,” said Nigel Bunnett, Ph.D., professor and chair of the Department of Molecular Pathobiology at NYU College of Dentistry. Bunnett led the study with Pierangelo Geppetti, MD, professor of clinical pharmacology at the University of Florence and director of the Headache Center of Careggi University Hospital.
While CGRP has been linked to migraine pain, the mechanism by which it causes pain has been a source of debate in the scientific community.
Nigel Bunnett
“The success of CGRP monoclonal antibodies for migraine and the inability of antibodies to penetrate the blood brain barrier suggest that CGRP causes pain in the periphery rather than within the brain,” Bunnett, an NYU Pain Research Center researcher, added.
Bunnett and his colleagues focused on Schwann cells, which are found outside the brain in the peripheral nervous system, to investigate the cellular mechanism of CGRP-evoked pain. The researchers looked at mice that had the CGRP receptor, known as CLR/RAMP1, disabled in Schwann cells. They altered the CGRP receptor by deleting RAMP1, one of the receptor’s two important components, from Schwann cells in the mouse’s face.
CGRP administration made the facial region of normal mice extremely sensitive, acting as a proxy for migraine pain. CGRP, on the other hand, did not cause pain in mice lacking the CGRP receptor in Schwann cells. In a subsequent experiment, the researchers used capsaicin, a chemical found in hot chili peppers. Capsaicin activates an ion channel called TRPV1, which causes pain by releasing CGRP. Again, capsaicin did not cause migraine-like pain in mice lacking the CGRP receptor in Schwann cells, indicating that the CGRP receptor in Schwann cells is important in migraine pain.
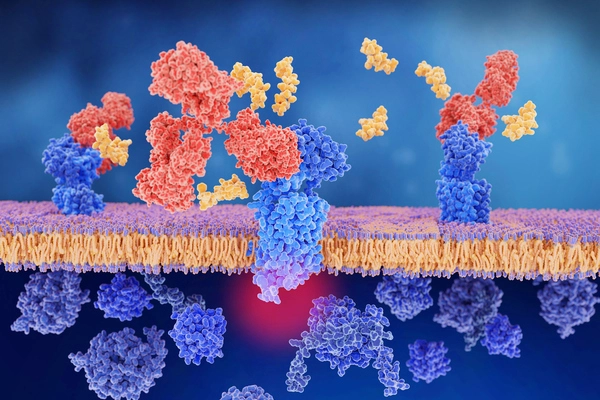
The researchers then discovered what happens within human Schwann cells to signal pain. When CGRP binds to its receptor on a Schwann cell, the receptor moves into a compartment within the cell known as an endosome. The CGRP receptor continues to signal for extended periods of time within endosomes. This signaling produces nitric oxide, a pain mediator, which is then released from the Schwann cell and interacts with an ion channel on an adjacent neuron called TRPA1. TRPA1 stimulates the neuron and sends pain signals.
This new understanding of how pain originates within Schwann cells provided the researchers with two treatment options for migraine pain: preventing the CGRP receptor from entering endosomes in the first place, or using nanoparticles to deliver drugs targeting the CGRP receptor in endosomes.
The researchers inhibited clathrin and dynamin, two proteins involved in the transport of substances into cells, to keep CGRP receptors out of endosomes. Clathrin and dynamin inhibition reduced pain signaling, suggesting a promising target for novel migraine treatments.
The researchers also successfully blocked CGRP-evoked pain using nanoparticles in which they encapsulated a small molecule drug that binds to and blocks the CGRP receptor. While most drugs only reach the surface of cells, nanoparticles can be designed to help medicine reach the endosome inside a cell and to release the drug once it arrives at the intended location. In Schwann cells, the nanoparticles carried the drug into endosomes and blocked the CGRP receptor, which strongly inhibited migraine pain.
“While the role of CGRP in migraine pain is well established, this is the first study to directly link Schwann cells to migraine pain. Based on our improved understanding of how pain is signaled from within endosomes, it offers potential new approaches to treating migraine” Bunnett stated.
The researchers are collaborating with several partners, including the National Institutes of Health’s National Center for Advancing Translational Sciences, to continue studying the use and safety of nanoparticle drug delivery before these treatments can be tested in humans.