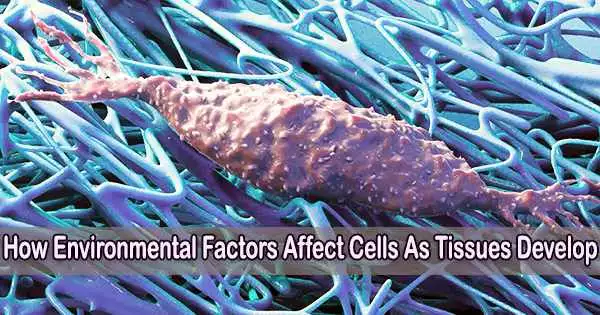
Cells and tissues grow through a process called cell proliferation and tissue remodeling. It is important to note that cell and tissue growth are tightly regulated processes in the body, ensuring that growth occurs in a controlled manner and maintains tissue homeostasis.
How does an embryo develop? How do children grow, wounds heal or cancer spread?
All of this has to do with the growth of body tissue. Understanding this evolution in depth is one of the main research focuses of ETH Professor Viola Vogel and her senior assistant Mario C. Benn. They have diverged from established scientific avenues in their pursuit.
Biology has long been the study of cells and the biochemistry of their metabolic activities, frequently independent of the cells’ environmental environments. The extracellular matrix (ECM), a fibrous framework that surrounds body cells, is the focus of Vogel and Benn, in contrast. This matrix, which is a crucial part of every tissue, is created by the cells themselves.
There are many different interactions between body cells and this fibrous matrix. Research in recent years has consistently demonstrated that not all of these interactions are solely biological. Some are actually mechanical or physical. Cells, for instance, can detect mechanical impulses from this extracellular matrix.
Vogel and Benn have now successfully replicated tissue growth in vitro and studied this process in depth with their team of experts.
“Our results underline the importance of the interactions between cells and the extracellular matrix,” Benn says. In time, he hopes to make medical use of these findings to prevent wound-healing disorders, for example, or in the therapy of cancer and connective-tissue diseases.
Cell transformation
Their study, now published in Science Advances, focused on two cell types: fibroblasts and myofibroblasts. Each of these can shift into the other and is essential for the functionality of human tissue. Our organs’ connective tissue contains fibroblasts, which are responsible for maintaining the extracellular matrix’s health and ensuring its constant renewal.
Our results underline the importance of the interactions between cells and the extracellular matrix.
Mario C. Benn
Myofibroblasts, which are crucial for wound healing and the development of new tissue, are formed when an injury occurs or tissue growth is necessary. In addition to making a lot of ECM, myofibroblasts are powerful enough, for instance, to bind tissue in wounds.
“When it comes to wound healing, myofibroblasts are our friends,” Benn says. Once their work is done, however, it is important that these myofibroblasts change back into the less active fibroblasts. If not, this can lead to fibrosis the excessive formation of scar tissue. Myofibroblasts are also found in cancer tissue. For many cancers, high levels of these cells is associated with a poor prognosis.
Three-dimensional matrix
The biochemical steps that occur when myofibroblasts transform into fibroblasts are understood to some extent. But there hasn’t been much investigation into how the ECM affects this cellular change.
“With conventional cell-culture methods, the cells grow flat across the culture dish. This leads to the formation of an unnaturally planar ECM,” Vogel explains. “And anyway, research up to now has generally ignored the ECM. But studying cells without the extracellular matrix is a bit like studying the behavior of spiders without their web.”
The method used by Vogel and Benn is quite different. The ETH researchers have improved it since it was first created at the Max Planck Institute of Colloids and Interfaces in Potsdam. They employ a silicone scaffold with microscopic triangular clefts that is coated with certain proteins and placed in a tissue culture medium.
In these clefts, new tissue and a more natural ECM emerge over the course of two weeks. Growth begins at the apex, progressively filling the cleft as the tissue grows.
The researchers noticed that myofibroblasts are consistently found right at the growth front, or in the region of the tissue that is just beginning to form. Additionally, they were able to demonstrate how myofibroblasts in this region create new ECM, first in a provisional form, then in a more stable form, before reverting to their original state as fibroblasts.
“The processes are similar to those that take place in human subcutaneous tissue during the late phase of wound healing,” Benn says.
The researchers were also able to demonstrate that one of the catalysts for the conversion of myofibroblasts to fibroblasts is a quickly altering ECM. Additionally, this reversion is facilitated by the transition from a stretched to a relaxed state of a specific type of ECM fiber called fibronectin. Similar interacting processes appear to take place during wound healing.
The researchers then used a variety of chemicals that alter the extracellular matrix’s composition or structure to purposefully interfere with the cell transition. Thus, scientists were able to mimic what happens in diseases like fibrosis or cancer, when myofibroblasts are maintained by the extracellular matrix rather than returning to fibroblasts as in healthy tissue.
Future mechano-medicine
The scientists are hoping that using these tiny tissue cultures they might learn more about how human cells interact with their extracellular matrix. This will not only do away with the frequently required use of animal testing in biomedical research, but it will also allow for the testing of potential drug candidates in the future.
“These applications and research questions are low-hanging fruit,” Benn explains. “If we can understand how myofibroblasts and fibroblasts change into one another, and control that process, then we can also make major progress with conditions such as wound-healing disorders, fibrosis and cancer.”
Benn and Vogel also refer to a future field called mechano-medicine. This term describes the medical application of findings from the field of mechanobiology: the study of how cells can sense and process mechanical signals. In other words, the goal of mechano-medicine is to integrate the knowledge from mechanobiology into the practice of medicine.
The researchers intend to eventually apply mechano-medicine to the creation of fresh diagnostic techniques for the early recognition of fibrotic tissue.
“With many conditions, including pulmonary fibrosis, successful treatment depends on early detection,” Benn says.
Myofibroblasts cannot be reliably identified in lung tissue using current screening techniques. Benn is now hoping that additional research on the extracellular matrix may uncover biomarkers that make it possible to detect fibrosis and other connective-tissue illnesses more quickly and easily.