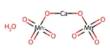
Fluorellestadite is a rare nesosilicate of calcium, with sulfate and fluorine, with the chemical formula Ca10(SiO4)3(SO4)3F2. It is a rare nesosilicate of calcium, with sulfate and fluorine, belonging to the apatite group and forming a series with hydroxylellestadite. It is a member of the apatite group and forms a series with hydroxylellestadite.
It is a rare mineral that occurs in coal dumps in burned fragments of petrified wood at its type locality in Kopeisk in the Urals region of Russia.
General Information
- Category: Nesosilicates (Apatite structural group)
- Formula: Ca10(SiO4)3(SO4)3F2
- Crystal system: Hexagonal
- Crystal class: Dipyramidal (6/m)

Fig: Fluorellestadite
Properties
Fluorellestadite shows imperfect cleavage perpendicular to the long crystal axis. The mineral is very brittle and breaks with a conchoidal fracture. Its hardness is 4.5, between that of fluorite and apatite, and its specific gravity is 3.03 to 3.07, similar to that of fluorite. It is easily soluble in dilute hydrochloric and nitric acids and is not radioactive.
- Formula mass: 503.55 g/mol
- Color: Light red, yellow, bluish green or colorless
- Crystal habit: Acicular or hexagonal prismatic crystals, and fine-grained aggregates
- Fracture: Conchoidal
- Tenacity: Very brittle
- Mohs scale hardness: 4.5
- Luster: Sub-resinous to vitreous
- Streak: White with a weak bluish tint
- Diaphaneity: Transparent to translucent
- Specific gravity: 3.03 to 3.07
Occurrence and associations
The type locality is Coal Mine No. 44, Kopeisk, Chelyabinsk coal basin, Chelyabinsk Oblast, Southern Urals, Russia, and type material is held at the Fersman Mineralogical Museum, Academy of Sciences, Moscow, Russia. Ellestadite is a skarn mineral.
It occurs associated with diopside, wollastonite, idocrase, monticellite, okenite, vesuvianite, calcite and others at Crestmore, Riverside County, California, US. At Crestmore a contact zone exists between crystalline limestone and granodiorite.
Information Source: