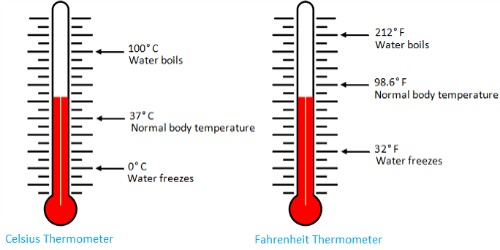
The boiling point of a substance is the temperature at which the substance boils, or enters a state of rapid evaporation. It is the temperature at which a liquid changes to a gas (vapor) at normal atmospheric pressure. For pure water, this is 100° Celsius or 212° Fahrenheit. This is measured at one atmosphere, that is, the air pressure at sea level. The normal boiling point is the temperature at which the liquid boils when the external pressure is one atmosphere (760 torrs = 760 mm Hg = 1 atm = 101.3 kPa = 14.7 psi). At the boiling point molecules anywhere in the liquid may be vaporized.
Boiling points can be changed in several ways. The addition of solutes or other substances usually changes the boiling point. Additionally, changing the pressure on a liquid changes its boiling point. The boiling point becomes lower as the external pressure is reduced. As an example, at sea level, the boiling point of water is 100 C (212 F), but at 6,600 feet the boiling point is 93.4 C (200.1 F).
The boiling point of a liquid depends on the pressure of the surrounding air. Once the liquid starts to boil, the temperature remains constant until all of the liquid has been converted to a gas. An increase in air pressure increases the boiling point; a decrease decreases the boiling point. In the low-pressure environment at the top of Mt Everest for example, water boils at only 69 °C. (156.2 °F). It can also be defined in terms of vapor pressure as the temperature at which the vapor pressure of the liquid becomes equal to atmospheric pressure. For example, the normal boiling point of water is 100oC. But if you try to cook an egg in boiling water while camping in the Rocky Mountains at an elevation of 10,000 feet, you will find that it takes longer for the egg to cook because water boils at only 90oC at this elevation. The boiling point of a liquid is lowered if the pressure of the surrounding gases is decreased.
Sugar, salt or other non-volatile solutes in water will usually make the boiling point higher. Alcohol, in contrast, is a volatile chemical that lowers the boiling point of water. Even a large amount dissolved in the water will usually make only small changes in the boiling point.