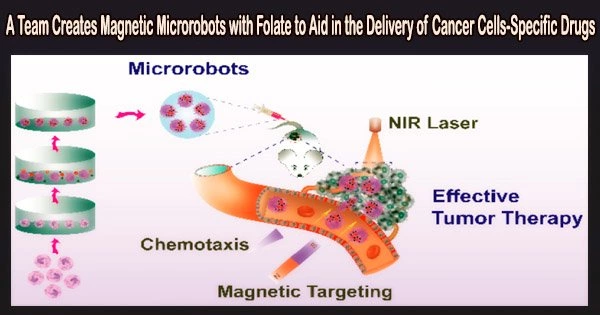
Microrobots’ limited capacity to help medications enter cells reduces the effectiveness of their therapeutic effects. Folic acid (FA), a cancer-targeting chemical, has been added to microrobots by a research team, as reported in Cyborg and Bionic Systems, to facilitate drug uptake by cancer cells through receptor-ligand-mediated endocytosis.
As a result, a drug delivery system is created that can use magnetic fields to pinpoint lesion locations and endocytose loaded medications into the cytoplasm.
Untethered microrobots have made outstanding progress in a number of areas, including tissue engineering, drug delivery, environmental cleanup, and minimally invasive surgery. Due to its high temporal and spatial control, deeper tissue penetration, and exceptional biosafety, magnetic field actuation is a commonly utilized technique.
However, practical issues occur when drug-delivery microrobots may only be able to reach the cells’ immediate vicinity without helping the medications enter the cells. Due to the possibility that the medications may not enter the cells where they are needed, this restriction may diminish the efficiency of the treatment.
Microrobots should be capable of accurate targeting in order to increase the efficacy of therapeutic use. To improve a microrobot’s ability to target specific cancer cells and cells’ ability to absorb drugs, researchers added folic acid (FA).
The ability of FA to facilitate the absorption of medicines by cancer cells through receptor-ligand-mediated endocytosis is made possible by the selective overexpression of the FR on the surface of cancer cells.
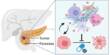
The developed magnetic microrobot system consists of biodegradable gelatin methacryloyl (GelMA)-based ABF microhelix and FA-loaded Fe@ZIF-8 (MOF) nanoparticles. For the purpose of treating cancer, therapeutic medications like DOX can be put into the hydrogel network of the microrobots. The microrobot can be guided and fixed at the lesion site with the help of an external rotating magnetic field, ensuring that therapeutic medications accumulate near cells.
Endocytosis, which results in the entry of MOF(FA) and DOX into the cells, can occur when the entire binding of FA on the microrobots and FR on the surface of cancer cells occurs. The interaction of receptors and ligands is improved and therapeutic effectiveness is increased by the aggregation of microrobots close to cells.
According to the study’s findings, microrobots with FA exhibited more pronounced cell inhibition than those without FA. As a result, the ABF-MOF(FA) drug delivery system, which combines magnetic manipulation and active FA targeting, has a promising future in the treatment of cancer.
The microrobot based on GelMA appeared to have a particular drug encapsulation ability and drug release ability in tests of the drug encapsulation capability, according to the release curve. The target ability of FA was proven by the MTT assay and the live/dead staining experiments.
Furthermore, tests on motion control and cells showed that microrobots can be precisely controlled by a magnetic field and carry out certain tasks. Finally, within 24 hours of being guided to the target region by a magnetic field, folate-loaded magnetic microrobots exhibited a clear anti-cancer impact.
The folate-targeted magnetic microrobot system’s large loading capacity, controlled navigation, and capacity to improve cancer cell targeting and inhibition make it an excellent candidate for cancer treatment.