Rice is a staple meal in many African countries, and viruses can cause substantial damage to rice harvests, reducing productivity and quality. Researchers frequently use a variety of strategies, including traditional breeding and genetic engineering, to create rice cultivars that are more disease-resistant.
Researchers at Mount Sinai’s Icahn School of Medicine have discovered an allergy route that, when inhibited, induces antitumor immunity in animal models of non-small cell lung cancer (NSCL).
In early parallel research in humans, combining immunotherapy with dupilumab, an Interleukin-4 (IL-4) receptor-blocking antibody often used to treat allergies and asthma, improved patients’ immune systems, with one out of six having a significant tumor decrease.
The findings were described in the issue of Nature.
Using single-cell technologies, we discovered that the immune cells infiltrating lung cancers, as well as other cancers we studied, exhibited characteristics of a ‘type 2’ immune response, which is commonly associated with allergic conditions like eczema and asthma.
Nelson LaMarche
“Immunotherapy using checkpoint blockade has revolutionized treatment for non-small cell lung cancer, the most common form of lung cancer, but currently only about a third of patients respond to it alone, and in most patients, the benefit is temporary,” says senior study author Miriam Merad, MD, PhD, Director of the Marc and Jennifer Lipschultz Precision Immunology Institute and Chair of the Department of Immunology and Immunotherapy at the Icahn School of Medicine at Mount Sinai. “A big focus of our program TARGET is to use single-cell technology and artificial intelligence to identify molecular immune programs that can dampen tumor immune response to checkpoint blockade.”
Checkpoint blocking, also known as a PD1 inhibitor, is a kind of cancer immunotherapy that allows T cells to release their cancer-killing potential.
“Using single-cell technologies, we discovered that the immune cells infiltrating lung cancers, as well as other cancers we studied, exhibited characteristics of a ‘type 2’ immune response, which is commonly associated with allergic conditions like eczema and asthma,” says Nelson LaMarche, Ph.D., a postdoctoral research fellow in Dr. Merad’s lab.
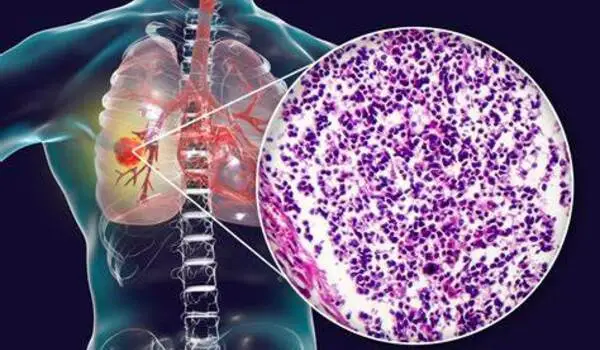
“These results led us to explore whether we could repurpose a medication typically used for allergic conditions to ‘rescue’ or enhance tumor response to checkpoint blockade,” says Thomas Marron, MD, PhD, Director of the Early Phase Trial Unit at Mount Sinai’s Tisch Cancer Center, and co-senior author of the study. “Strikingly, we found that IL-4 blockade enhanced lung cancer response to checkpoint blockade in mice and in six lung cancer patients with treatment-resistant disease. In fact, one patient whose lung cancer was growing despite checkpoint blockade had nearly all their cancer disappear after receiving just three doses of the allergy medication, and his cancer remains controlled today, over 17 months later.”
The researchers are encouraged by the preliminary findings, but they underline the need for larger clinical trials to confirm the drug’s usefulness in treating NSCLC. Beyond the clinical trial findings reported in the current Nature paper, the investigators have expanded the trial by adding dupilumab to checkpoint blockade for a larger group of lung cancer patients, and Dr. Marron recently received a grant from the Cancer Research Institute to study the effects in early-stage lung cancer as well. These trials are looking for biomarkers that can predict which cancer patients will benefit from dupilumab treatment and which will not.
“In our never-ending quest for progress, the Cancer Research Institute (CRI) gladly supports the imaginative team at Mount Sinai’s Icahn School of Medicine. Their findings support our commitment to sponsoring research across the whole discovery continuum, from lab to clinical implementation, using cutting-edge technology and data. We’re excited to see how our help is bringing new hope by identifying routes to improve checkpoint blockade responses. We support this discovery and are proud to be a part of its path from lab to clinic, underscoring our dedication to altering lives,” says Jill O’Donnell-Tormey, PhD, CEO and director of scientific affairs at CRI.