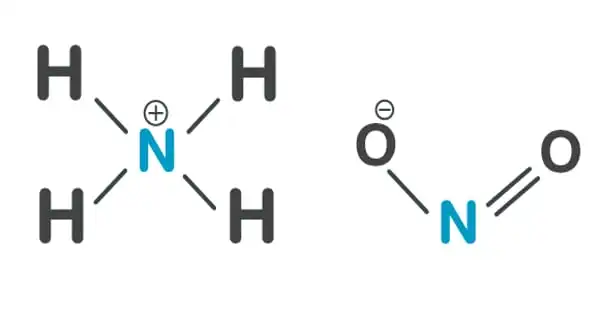
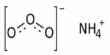The ammonium salt of nitrous acid is ammonium nitrite, NH4NO2. Because it is exceedingly unstable, it is not used in its pure form. It is not used in its pure isolated form since it is exceedingly unstable and decomposes even at ambient temperature into water and nitrogen. It decomposes to nitrogen and water at ambient temperature.
Ammonium nitrite is generated naturally in the atmosphere and can be made by absorbing equal parts nitric oxide and nitrogen dioxide in liquid ammonia. It is also produced by oxidizing ammonia with hydrogen peroxide or ozone. It can also be made by combining lead nitrite or barium with ammonium sulfate, potassium nitrite with ammonium perchlorate, or ammonium chloride with silver nitrite. The crystals obtained are colorless and dissolve in water.
Properties
Ammonium nitrite can burst at temperatures ranging from 60 to 70 °C, and it decomposes faster in a concentrated aqueous solution than in the form of a dry crystal. Because it is exceedingly unstable, it is not used in its pure form.
- Molecular weight: 64.06
- Density: 1.69 g/cm3
- Appearance: Pale yellow
- Melting point: Decomposes

Preparation
Ammonium nitrite is formed naturally in the air and can be produced by absorbing equal parts nitrogen dioxide and nitric oxide in aqueous ammonia. It can also be made by oxidizing ammonia with ozone or hydrogen peroxide, or by precipitating barium or lead nitrite with ammonium sulfate, silver nitrite with ammonium chloride, or ammonium perchlorate with potassium nitrite. The precipitate is filtered out, and the solution is concentrated. It crystallizes as colorless crystals that dissolve in water.
Ammonium nitrite can be produced by a reaction between silver nitrite and ammonium chloride, which also yields silver chloride.
2AgNO2 + NH4Cl = NH4NO2 + 2AgCl
Nitrous acid also reacts with ammonium hydroxide to produce ammonium nitrite and water.
HNO2 + NH4OH = NH4NO2 + H2O
This inorganic chemical compound explodes if it is heated above 60 to 70 °C. The rate of decomposition is faster when dissolved in concentrated solution when compared to in dry crystal form.
Chemical properties
Ammonium nitrite can explode at 60–70 °C and decomposes faster when dissolved in a concentrated aqueous solution than when in the form of a dry crystal. The chemical decomposes into water and nitrogen even at ambient temperature.
NH4NO2 → N2 + 2 H2O
When heated or exposed to acid, it decomposes into water and nitrogen. Ammonium nitrite solution is stable at higher pH and lower temperature. Any reduction in pH below 7.0 may result in an explosion because nitrite can react to it. A safe pH can be maintained by adding an ammonia solution. The mole ratio of ammonium nitrite to ammonia must be greater than ten percent.
Uses
Ammonium nitrite can be used as a microbiocide, rodenticide, and crop pesticide. It can also be used to synthesize nitrogen gas and ammonium cobalt-nitrite. However, it is most typically used in the manufacture of explosives.
Safety Hazards
The chemical compound ammonium nitrite is extremely harmful to both humans and aquatic animals. If we use it recklessly, it can produce a large explosion. If we heat the ammonium nitrite, it will be able to produce deadly vapors of ammonia and nitrogen oxides at the same time. Humans and aquatic animals are very hazardous to the chemical. It has the potential to produce an explosion. Heating it can produce toxic fumes of ammonia and nitrogen oxides.