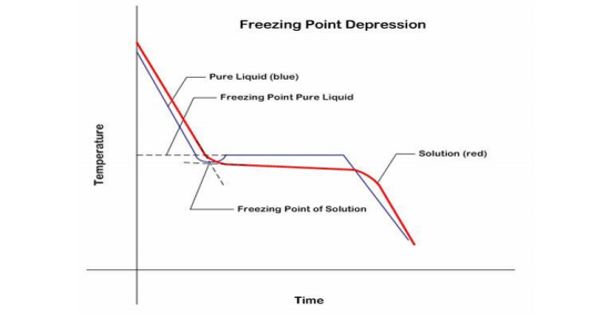
The melting points of compounds may be lower than the reported values because they may contain small amounts of impurities or solvents. Melting-point depression is the phenomenon of reduction of the melting point of material with a reduction of its size. A melting point is a useful indicator of purity as there is a general lowering and broadening of the melting range as impurities increase. This phenomenon is very prominent in nanoscale materials, which melt at temperatures hundreds of degrees lower than bulk materials. A substance (solid) containing soluble impurities usually melts at a lower temperature than the pure compound. It can also melt over a wide range of temperatures and is called the “melting point depression.”
Introduction
A melting point is a characteristic physical property of a substance. The melting temperature of bulk material is not dependent on its size. So melting point analysis is one of the simplest and most useful techniques for the identification of a chemical substance. However, as the dimensions of a material decrease towards the atomic scale, the melting temperature scales with the material dimensions. In general, the smaller the range of melting temperatures, the higher the purity of the sample. The decrease in melting temperature can be on the order of tens to hundreds of degrees for metals with nanometer dimensions.
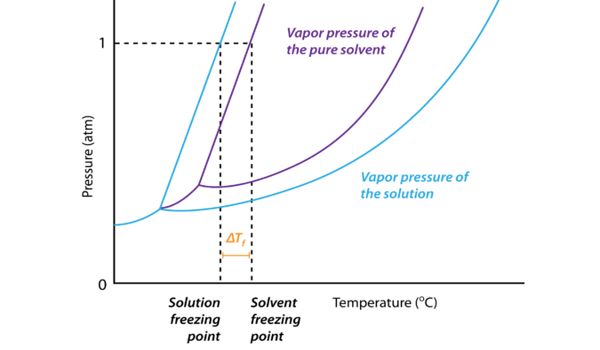
Melting-point depression is most evident in nanowires, nanotubes, and nanoparticles, which all melt at lower temperatures than bulk amounts of the same material. It’s not difficult to understand how impurities affect the melting point. Changes in melting point occur because nanoscale materials have a much larger surface-to-volume ratio than bulk materials, drastically altering their thermodynamic and thermal properties. Foreign substances in a crystalline solid disrupt the repeating pattern of forces that hold the solid together. So a smaller amount of energy is required to meet the part of the solid surrounding the impurity.
This article focuses on nanoparticles because researchers have compiled a large amount of size-dependent melting data for near-spherical nanoparticles. If less energy is required, then this explains the melting point depression (lowering) observed from impure solids. Nanoparticles are the easiest to study due to their ease of fabrication and simplified conditions for theoretical modeling. The more impure the solid is, the more the structure is disrupted and the greater the variation in intermolecular forces in different areas of the solid. The melting temperature of a nanoparticle decreases sharply as the particle reaches critical diameter, usually < 50 nm for common engineering metals.
Melting-point depression by soluble impurities is an entropy-driven phenomenon. Melting point depression is a very important issue for applications involving nanoparticles, as it decreases the functional range of the solid phase. The melting temperature is lowered compared to the pure solid, and the solid melts over a wider range of temperatures. Nanoparticles are currently used or proposed for prominent roles in catalyst, sensor, medicinal, optical, magnetic, thermal, electronic, and alternative energy applications. Nanoparticles must be in the solid-state to function at elevated temperatures in several of these applications.
Information Source: