Fluoroboric acid, also known as “hydrogen fluoroboric acid,” or tetrafluoroboric acid (archaically, fluoboric acid), is an inorganic compound with the chemical formula (H+)(BF4–), where H+ represents the solvated proton. It has the appearance of a colorless, odorless toxic liquid. Metals and tissue are corroded. It’s used in electroplating, metal washing, and the production of diazo salts, among other things. The molecular weight of this compound is 87.81. It’s a clear, colorless liquid that’s poisonous and corrosive, and it can’t be kept in a glass container for long.
Fluoroboric acid has a boiling point of 130°C and decomposes slowly. The solution was highly acidic, although it does not corrode glass at room temperature. It was dissolved in water and ethanol, thermal decomposition with water to produce oxygen fluorine boric acid and boron trioxide (may be concentrated to 30%).
H3OBF4 (oxonium tetrafluoroborate) can be expressed in water, but more realistically, many water molecules solvate the proton: (H(H2O)n+)(BF4–). Fluoroboric acid is a strong acid that only occurs in solution, with potassium tetrafluoroborate being the most common fluoroborate. Since MMFF94s unsupported element, MMFF94s unsupported atom valence, mixture, or salt, conformer generation is forbidden. Commercially accessible ethyl ether solvate is (H(Et2O)n+)(BF4–), where n is most likely 2. Unlike strong acids such as H2SO4 or HClO4, there is no pure unsolvated material.
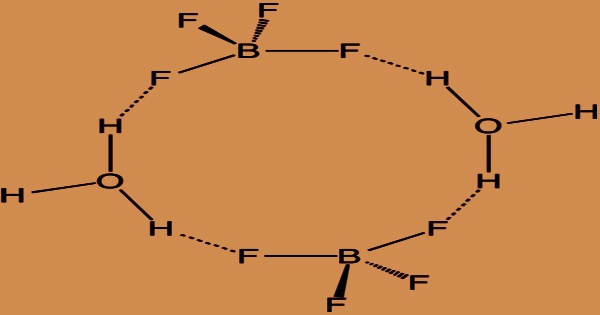
Fluoroboric acid is corrosive to the skin and mucous membranes, as well as irritant to the eyes and respiratory system. It can stifle fermentation even when diluted quite thinly. Maximum allowable concentration is 2.5 mg/m3. If splashed in the eyes or on the body, flush with water and thoroughly wash contaminated body parts with soap and water. It’s commercially available as a solution in water and other solvents including diethyl ether. It’s a heavy acid with a non-oxidizing conjugate base that’s weakly coordinating. It is structurally similar to perchloric acid, but it does not pose the same risks as oxidants.
Fluoroboric acid releases poisonous boron and hydrogen fluoride vapors when heated to decomposition. Pure HBF4 has been described as a “nonexistent compound,” as a sufficiently ‘naked’ proton is expected to abstract a fluoride from the tetrafluoroborate ion to give hydrogen fluoride and boron trifluoride:
(H+)(BF4–) → HF + BF3
Fluoboric acid is a stable chemical compound that is highly reactive with strong bases and cyanides. In the presence of steel, aluminum, zinc, and copper, it is extremely corrosive; in the presence of glass and stainless steel, it is extremely corrosive. A solution of BF3 in HF, on the other hand, is extremely acidic, with an estimated speciation of (H2F+)(BF4–) and a Hammett acidity function of –16.6 at 7 mol% BF3, easily qualifying as a superacid. Despite the fact that the solvent-free HBF4 has yet to be isolated, its solvates have been thoroughly studied.
Fluoroboric acid is only stable as a solvated ion pair, such as H3O+BF4–, and does not exist as a free, pure product. The acidity of fluoroboric acid is complicated by the fact that the term refers to several different species, each with a different acidity: H(OEt2)+BF4–, H3O+BF4–, and HF.BF3-. The hydrofluoric acid method involves slowly adding boric acid to hydrofluoric acid while stirring, keeping the reaction temperature below 40 °C, stopping mechanical stirring until the heating is full, leaving the mixture at room temperature for more than 2 hours, then filtering and purifying to obtain fluoroborate finished product.
4HF + H3BO3 → HBF3OH + HF + 2H2O
HBF3OH + HF → HBF4 + H2O
The fluoroborate ion is hydrolyzed in a calcium chloride solution and analyzed gravimetrically with the help of nitron. It’s used in light metal smelting, electroplating, and other processes. Fermentation inhibitors can be used in even very dilute solutions; they’re also used to make diazonium salts and acetal catalysts, which are used to determine sodium in the presence of magnesium and potassium ions. Aniline reacts with sodium nitrite as a raw material in concentrated hydrochloric acid to produce diazonium chloride, and then reacts with fluoroboric acid at a low temperature (-10 °C) to produce fluorophenyl.
Inorganic salts are used as intermediates in the production of flame-retardant materials and glazing frits, as well as in the electrolysis of boron. Skin and eyes can be severely burned if they come into contact with molten substance. Avoid any skin contact. Fluoroboric acid is used to remove metal surface oxide, silicate film, and as a corrosive before electroplating aluminum and alloys. 2.5 percent solution is used for electrolytic polishing of pure aluminum, as a rewashing lotion to remove flux and electroplating sections from the metal substrate, as a catalyst for alkylation and polymerization, preservatives, chemical reagents, and as the starting materials for various fluoborates. It’s also used in the printed wiring board industry and for the dissolution of sponge titanium and its alloys.
Information Sources: