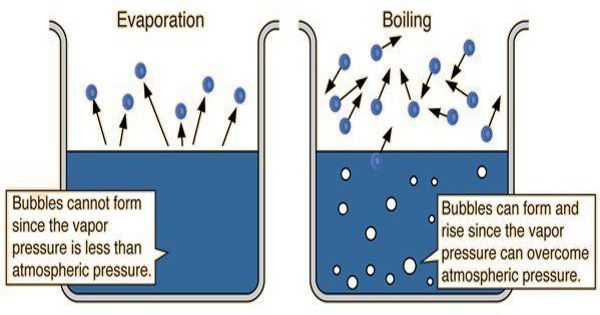
Vaporization is a process of phase transition in which a substance, changes its state from liquid to vapor. It can take place in two ways, i.e. evaporation and boiling. Boiling occurs when the temperature of the liquid is greater than the boiling point of the substance. Evaporation can occur at any temperature.
Evaporation is the process where liquid gets converted into vapour. The process of evaporation involves phase transition at a temperature below the boiling temperature. On the other hand, boiling of a substance takes place at boiling point, which may vary with the change in the environmental pressure. It refers to the process of heating up a liquid where the temperature of the liquid being heated up is greater than the boiling point of the substance.
Boiling is a process in which a substance changes its state from the liquid state to the gaseous state. Evaporation is a process in which a substance changes its state from the liquid state to the gaseous state without boiling.
Difference between Boiling and Evaporation –
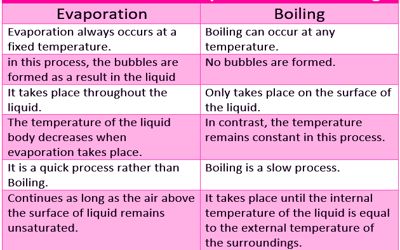
BOILING
- Boiling implies a vaporization process that turns liquid into gas when continuously heated. It is an unnatural process where the liquid gets heated up and vaporized due to continuous heating of the liquid.
- Boiling is a physical change and a type of rapid vaporization in which the liquid is converted to vapor when it is constantly heated at such a temperature that the vapor pressure of the liquid is the same as the external pressure, exerted by the surrounding.
- Boiling refers to the process of vaporization, wherein the liquid state is turned into the gaseous state, at a definite boiling point.
- The temperature at which boiling starts is known as the boiling point. It depends on the pressure exerted upon the liquid, i.e. the greater the pressure, the higher will be the boiling point.
EVAPORATION
- Evaporation is a natural process, wherein the liquid changes its form to gas due to the increase in temperature or pressure. It is a normal process that occurs when the liquid form changes into the gaseous form; while causing an increase in the pressure or temperature.
- The process can be used to separate the solid dissolved in the liquid, such as salt dissolved in water. It is a surface phenomenon, i.e. it happens from the surface of the liquid into vapour.
- Evaporation is defined as a natural process, in which the increase in temperature and/or pressure changes liquid into gas.
- Heat energy is the basic requirement for evaporation to take place, i.e. to split the bonds that hold the molecules of water together. In this way, it helps water to evaporate slowly, at the freezing point.
Boiling of a liquid occurs only at the boiling point of that liquid, i.e. it takes place only at a definite temperature. As against, evaporation process can occur at any temperature. Water boils at 100°C, and the temperature does not rise even if heat is continuously supplied to it. As against, the rate of evaporation depends on the surface area, in the sense that the larger the area, the faster would be the process. In boiling, the temperature of liquid remains same, whereas in case of evaporation the temperature of the liquid tends to decline.