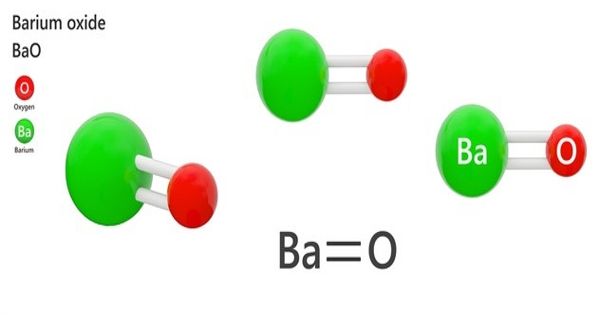
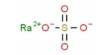
Barium oxide (BaO) is a white or yellowish-white poisonous heavy powder used esp as a dehydrating agent. It is a white hygroscopic non-flammable compound. It has a cubic structure and is used in cathode ray tubes, crown glass, and catalysts. It is widely used as a drying agent for solvents, in cathode ray tubes, catalysts, and crown glass. It is harmful to human skin and if swallowed in large quantity causes irritation. Excessive quantities of barium oxide may lead to death.
It is prepared by heating barium carbonate with coke, carbon black or tar or by thermal decomposition of barium nitrate. On reacting with water it generates heat that increases the fumes concentration in the air.
Properties
Barium oxide appears as a white to yellow powder. It is a white ionic crystal that has a non-flammable nature. It is a cubic structured compound that can be prepared by different methodologies, such as thermal decomposition of barium nitrate, and by heating barium carbonate with carbon black or tar.
- Molecular Weight: 153.326
- Appearance: White to very pale yellow crystalline solid
- Melting Point: 1,923° C (3,493° F)
- Boiling Point: ~2000 °C (3,632° F)
- Density: 5720 kg/m-3
- Solubility in H2O: N/A
- Exact Mass: 153.9 g/mo
- Monoisotopic Mass: 153.900157 Da

Preparation
Barium oxide is made by heating barium carbonate. It may also be prepared by thermal decomposition of barium nitrate. Likewise, it is often formed through the decomposition of other barium salts.
2Ba + O2 → 2BaO
BaCO3 → BaO + CO2
Barium oxide can be produced by thermally decomposing barium carbonate, BaCO3 or barium nitrate, Ba(NO3)2. The reactions are as follows:
BaCO2 → BaO + CO2
2Ba(NO3)2 → 2BaO + 4NO2 + O2
Uses
- Barium oxide is used to coat hot cathodes.
- It is used in the production of optical crown glass.
- It is used as a coating for hot cathodes, for example, those in cathode ray tubes. It replaced lead(II) oxide in the production of certain kinds of glass such as optical crown glass.
- It also has use as an ethoxylation catalyst in the reaction of ethylene oxide and alcohols, which takes place between 150 and 200°C.
- It is also a source of pure oxygen through heat fluctuation. It readily oxidises to BaO2 by formation of a peroxide ion.
Safety issues
Barium oxide is an irritant. It is a toxic and corrosive compound which is non-combustible and water sensitive. If it contacts the skin or the eyes or is inhaled it causes pain and redness. However, it is more dangerous when ingested. When this compound catches fire it liberates irritating, toxic, and corrosive gases. It can cause nausea and diarrhea, muscle paralysis, cardiac arrhythmia, and can cause death.
Information Source: