Irradiation is used in antibacterial photodynamic treatment to create reactive oxygen species that destroy germs. Only surface infections can be treated using this method since it needs external light and oxygen.
A Chinese research team has now described a molecular “singlet oxygen battery” that can be “charged” with reactive oxygen and then released in deep tissue layers to target methicillin-resistant staphylococcus. The discovery was published in the journal Angewandte Chemie.
Antibiotic-resistant bacteria are on the rise. Despite frequently being innocuous to healthy people, dreaded multidrug-resistant “hospital pathogens” like methicillin-resistant Staphylococcus aureus (MRSA) enter the body through wounds from recent surgery or wounds from injuries. They also infect immunocompromised patients. Because antibiotics are not effective, there is sometimes no remedy.
Antibacterial photodynamic treatment, which is currently commonly used in dentistry, is one incredibly promising alternative. In this method, a chemical that can be activated by light (a photosensitizer) is exposed to light, which starts a photodynamic process that results in singlet oxygen (1O2), an excited form of oxygen.
Unlike antibiotics, this substance simultaneously attacks multiple biomolecular sites on the bacteria. It is easy to use, safe, painless, and generally free of side effects.
Unfortunately, because the required light only reaches a few millimeters into the tissue, it has only been effective for surface infections. Deeper tissue layers do not receive enough oxygen either for treatment to be effective.
A team led by Bingran Yu and Fu-Jian Xu at Beijing University of Chemical Technology has now developed a new approach to photodynamic therapy: a “singlet oxygen battery” that can be used to fight deep bacterial infections because it requires neither light nor external oxygen.
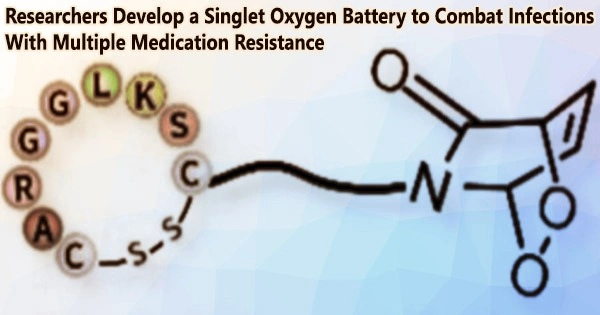
The first step is the irradiation-induced conversion of oxygen into reactive singlet oxygen in the presence of a light-absorbing molecule (photosensitizer). The “battery” is “charged” with the singlet oxygen. This “battery” is made up of pyridone, a unique nitrogen-containing six-membered carbon ring that firmly holds the singlet oxygen.
The reactive oxygen molecule bridges two opposite vertices of the ring (endoperoxide). The molecular batteries build up around and inside the MRSA bacteria and continuously release their singlet oxygen because a peptide attached to the ring uniquely “recognizes” MRSA bacteria.
Thus, multiple parts of the bacteria, including their membrane, DNA, enzymes, and other proteins, are concurrently attacked. This makes the development of resistance virtually impossible.
The singlet oxygen battery was proven to be extremely efficient in treating lung infections brought on by MRSA when nebulized into mice. Systemic side effects were not observed.