The creation of effective medications is only half the battle in the fight against cancer; it’s also critical to consider how medications can be administered to the body’s diseased areas effectively and securely.
The difficulty of drug distribution is particularly relevant for RNA therapies that target RIG-I, a crucial immuno-modulatory receptor. The receptor is capable of triggering immunological responses that can destroy cancer cells when activated by specific kinds of RNAs.
RNA-based medications must be packaged in suitable carriers because RNAs are naturally unstable and delicate. This will help to prevent degradation and encourage effective uptake by the target cancer tissues.
A study led by researchers at the Yong Loo Lin School of Medicine, National University of Singapore (NUS Medicine) in collaboration with the Lee Kong Chian School of Medicine, Nanyang Technology University, Singapore (LKCMedicine, NTU Singapore), and A*STAR’s Genome Institute of Singapore (GIS) demonstrated that nano-sized vesicles released by red blood cells are a viable platform for delivering immunotherapeutic RNA molecules to suppress breast cancer growth and metastasis.
Red blood cell extracellular vesicles (RBCEVs), which are tiny, lipid membrane-bound particles generated by red blood cells, were successfully used in the study to carry RIG-I-activating RNAs to slow the growth of cancer. This research was published in the Journal of Extracellular Vesicles.
In prior research, scientists had also found that these vesicles are perfect therapeutic carriers with a built-in capacity to deliver bioactive chemicals to a variety of cell types.
The promising results highlight two key strengths of this innovative platform the capacity for efficient delivery of different therapeutic cargoes, as well as the possibility for genetic modifications to enhance targeting to more cancer types.
Dr. Tam Wai Leong
Assistant Professor Minh Le from the Institute for Digital Medicine (WisDM) and Department of Pharmacology at NUS Medicine, who led the study, explained, “With the discovery of these vesicles’ ability to deliver therapeutics effectively to targeted receptors, we hope that our research can lead to better treatment outcomes for cancer patients. The correct homing of the therapeutics to diseased cells is also critical in minimizing off-target effects that can result in toxicity.”
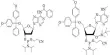
In order to activate the RIG-I pathway, cause cell death in breast cancer cell cultures, and decrease tumor growth in laboratory models of breast cancer, two new RNA molecules were created at LKCMedicine and packaged into RBCEVs.
Additionally, the scientists modified RBCEVs to increase their ability to specifically target lung-based metastatic cells.
Associate Professor Luo Dahai, Associate Professor of Infection and Immunity at LKCMedicine, said, “Asst Prof Le’s RBCEV technology can overcome several hurdles related to therapeutic RNA delivery and unleash the anticancer potential of our immunomodulatory RNA (immRNA). I am thrilled to see the success of our collaboration.”
Dr. Tam Wai Leong, Group Leader and Associate Director at GIS, one of the collaborators of the study, added, “The promising results highlight two key strengths of this innovative platform the capacity for efficient delivery of different therapeutic cargoes, as well as the possibility for genetic modifications to enhance targeting to more cancer types.”
In cooperation with the National University Cancer Institute and the Cancer Science Institute of Singapore, the team intends to carry out an additional study to further analyze the role of RBCEVs in delivering a wider spectrum of treatments to more cancer cell types.
Carmine Therapeutics, an EVX Ventures firm that aspires to create the next generation of gene therapy based on RBCEVs for the treatment of cancer and rare disorders, is actively researching RBCEV technologies at the same time.
“We hope to expand the therapeutic value of the RBCEV platform to more cancer types and increase the reach of such novel forms of therapy to benefit more cancer patients,” said Asst Prof Minh Le, who is also one of the co-founders of the company.