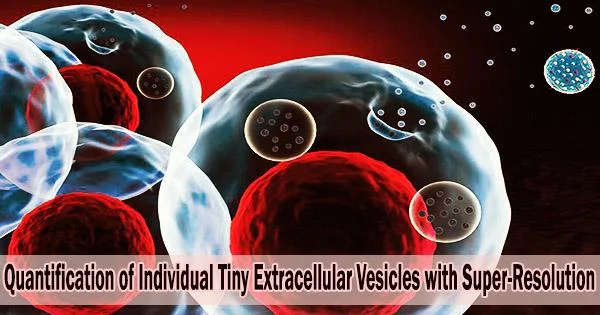
Tumorigenesis and cancer development are well acknowledged to be multistep processes. A complicated combination of imaging and invasive tissue biopsies forms the basis of the most often used method for cancer diagnosis and prognosis, which informs therapy choices. However, the methods are not always sensitive to early-stage cancer diagnosis.
The nanometer-sized, bilayer lipid carriers known as small extracellular vesicles (sEVs) carry a range of cargos, including lipids, proteins, metabolites, RNAs, and DNAs.
In practically all bodily fluids, sEVs released from the initial cancer cells can be found. As they represent the dynamic biological changes connected to the expanding tumors and identify the stages of cancer progression, they can become potential circulating biomarkers in liquid biopsies.
By expanding the resolution’s range toward nanoscale scales past the diffraction limit, super-resolution microscopy techniques have arisen.
Researchers from the University of Technique Sydney, under the direction of Professor Dayong Jin, have created a novel technology based on Lanthanide-doped EV-targeting Nanoscopic Signal-amplifiers (LENS). The research was recently published in the journal eLight.
Their paper, “Upconversion Nanoparticles for Super-resolution Quantification of Single Small Extracellular Vesicles,” has enormous potential in cancer diagnosis and prognosis.
Upconversion nanoparticles made in a lab have nonlinear photo-switchable characteristics. They allow for sub-30 nm optical resolution in a novel form of super-resolution nanoscopy.
In recent work, the researcher further improved ultra-sensitivity in the quantitative detection of sEVs using nanophotonic probes. These probes exhibited sensitivity improvements over the traditional enzyme-linked immunosorbent assay (ELISA) of roughly three orders of magnitude.
The surface biomarkers on individual EVs are super-resolved by the researchers’ further improvement in imaging resolution. Utilizing uniform, brilliant, and photostable nanophotonic probes is the basis of the method. Each one contains tens of thousands of lanthanide ions and is heavily doped.
The sEVs were first isolated in their experiment on a slide coated with CD9 antibody and sandwiched by an EpCAM antibody that had been biotinylated. The EpCAM antibody was later tagged with streptavidin functionalized upconversion nanoprobes for signal amplification. Single sEVs can be visualized using a super-resolution microscope and a doughnut-shaped laser beam thanks to the nanoprobes on them.
A single nanoprobe placed in the center of the doughnut beam produces an emission pattern that has a dip in it. The two neighboring nanoprobes can then be super-resolved in the nanoscale beyond the diffraction limit.
The researchers show that a library of upconversion nanoprobes doped with multiple types and variable concentrations of emitters may be used to accomplish super-resolution imaging of single sEVs. They confirm that tumor epitope epithelial cellular adhesion molecule (EpCAM) on both large EVs and single sEVs may be precisely targeted by antibody coated nanoprobes.
The researchers can count the precise number of nanoprobes on each sEV using super-resolution imaging. They have demonstrated that it is theoretically conceivable to evaluate the size and steric hindrance of nanoprobes on individual sEVs.