Other chemicals are emerging as major causes of stratospheric ozone depletion as the Earth’s ozone layer recovers from past emissions of now-banned CFCs and halons. Scientists in the atmosphere have been looking for the sources of about one-third of the major threats, methyl bromide, and methyl chloride. According to new research, common copper-based compounds produce these compounds when they interact with soil and seawater, with sunlight increasing production by a factor of ten.
According to a new study from the University of California, Berkeley, copper released into the environment from fungicides, brake pads, antifouling paints on boats, and other sources may be significantly contributing to stratospheric ozone depletion.
UC Berkeley geochemists show in a paper published in the journal Nature Communications that copper in soil and seawater acts as a catalyst to convert organic matter into methyl bromide and methyl chloride, two potent halocarbon compounds that deplete ozone. The situation is exacerbated by sunlight, which increases the production of these methyl halides by a factor of ten.
The findings help to explain, at least in part, the origin of much of the methyl bromide and methyl chloride in the stratosphere. Since the 1989 global ban on CFC refrigerants and brominated halons used in fire extinguishers, these methyl halides have become the new dominant sources of ozone-depleting bromine and chlorine in the stratosphere. As the long-lived CFCs and halons slowly disappear from the atmosphere, the role of methyl halides increases.
“If we don’t know where methyl bromide and methyl chloride are coming from, then how can we make sure that those compounds are reduced along with CFCs?” said the paper’s senior author, Robert Rhew, UC Berkeley professor of geography and of environmental science, policy and management. “By 2050, we should be back to relatively normal ozone, but things like the continued emissions of methyl bromide and methyl chloride are road bumps in the road to recovery. Copper usage in the environment is projected to increase rapidly in the next few years, and this should be considered when predicting future halogen load and ozone recovery.”
We did a back-of-the-envelope calculation to see what impact copper sulfate would have and estimated that it could be responsible for 4.1 gigatonnes of methyl bromide per year, which is about 10% of the missing source.
Robert Rhew
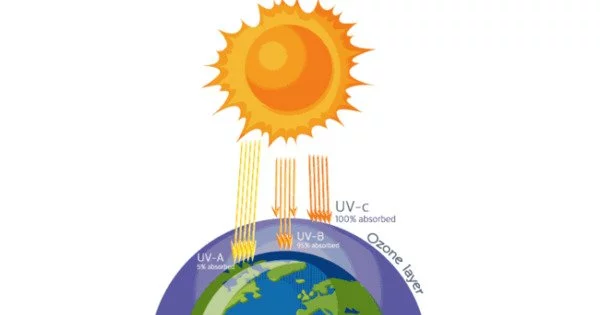
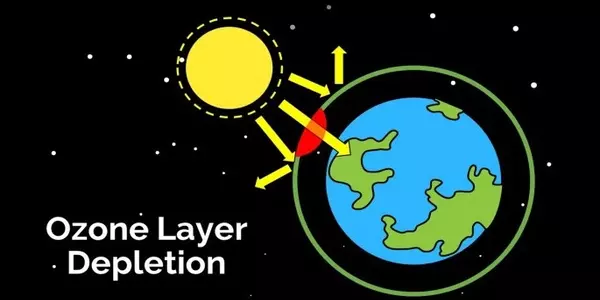
The ozone layer protects us from cancer-causing ultraviolet light from the sun, but chemicals containing chlorine and bromine, such as CFCs and halons, were discovered in the 1980s to destroy the ozone layer, creating thinner layers in the stratosphere that let in more of the dangerous radiation. Despite a ban on the production of CFCs and halons, the major sources of halogens, the ozone layer has yet to repair itself. According to Rhew, the ozone hole over Antarctica was at its worst last year.
The persistence of the ozone hole is primarily due to the persistence of banned ozone-depleting compounds, which take decades to dissipate in the stratosphere. However, some ozone-depleting chemicals are still being emitted. Even some replacements for banned refrigerants are coming under scrutiny.
Among the major contributors today are methyl chloride and methyl bromide. One atom of bromine is 50 times more destructive to ozone than one atom of chlorine.
“We’ve banned methyl bromide, but are other changes that we’re making in the environment causing large emissions of this compound into the atmosphere? With the increase in the use of copper, it appears that copper-catalyzed production is an increasing source, as well,” Rhew said.
Yi Jiao, the first author and a former UC Berkeley doctoral student who is now a postdoctoral fellow at the University of Copenhagen in Denmark, noted that copper compounds are allowed on organic crops, a legacy of its use in farming since the 1700s, including as a major antifungal agent in the Bourdeax mixture used in France since the 1880s to prevent downy mildew on grapes. Because of this history, copper contamination of soils is a major issue in Europe today. Copper’s ozone-depleting ability is another source of concern, according to the authors.
“Please note that organic agriculture is not a major cause for ozone depletion. However, copper-based fungicides appear to have atmospheric side effects that might be considered in terms of overall environmental impact,” Jiao tweeted this week. “With widespread use of copper in the environment, this potentially growing impact should be considered when predicting future halogen load and ozone recovery.”
Copper + soil + sunlight = methyl halides
The connection between copper and methyl halides was first revealed through a series of research projects conducted by UC Berkeley undergraduate researchers. Rhew asked them to test the impact of metal ions, starting by replicating previously published work on iron in soils. When this produced small amounts of methyl halides, Rhew then asked them to investigate a different metal – copper – in the form of copper sulfate, one of the most common copper compounds used today.
“We replicated the iron experiment and then thought, ‘Let’s look at a different transition metal, like copper, and see if it has a similar effect,'” Rhew said. “When we added copper sulfate to soil, it produced a tremendous amount of methyl halides, and this surprised us. And then another undergraduate did the experiment with seawater, and that produced an impressive amount of methyl halides, as well. So, we knew there was a novel process going on, but we only had a few pieces to the puzzle until Yi conducted a suite of creative experiments to put it all together.”
Jiao and Rhew devised more in-depth experiments, collecting soil samples from the Oxford Tract, an agricultural research plot near the UC Berkeley campus, and subjecting them to various treatments that included varying amounts of copper and oxidants. While copper alone produced some methyl bromide and methyl chloride in soil and seawater, the addition of sunlight and/or hydrogen peroxide (which is produced in soil by microbes or sunlight) produced more than five times the amount of methyl halides and extended copper activity from about a week to between two and three weeks.
Yi sterilized the soil, which increased the amount of methyl halide produced even more. However, after removing all organic material, soil incubated with copper produced no methyl halides. This led him to concentrate on chemicals such as catechol and guaiacol, which are frequently used as proxies for soil organic carbon because they both have a phenol ring structure similar to that of organic matter.
Increasing the concentrations of copper sulfate or hydrogen peroxide in catechol-halide solutions increased methyl halide emissions, whereas emissions were near zero when any of these substrates was absent. Yi discovered that sunlight functioned similarly to hydrogen peroxide in increasing methyl halide production. Exposing copper-amended solutions to sunlight increased emissions fourfold in seawater.
The researchers suspect that one common form of copper ion, Cu(II), is oxidizing organic material to liberate methyl radicals, which readily combine with chlorine and other halogens in the soil or seawater to form methyl halides. Both sunlight and hydrogen peroxide subsequently reoxidize the copper — from its cuprous (I) to cupric (II) state — so that it can act again and again to generate more methyl halides.
“We did a back-of-the-envelope calculation to see what impact copper sulfate would have and estimated that it could be responsible for 4.1 gigatonnes of methyl bromide per year, which is about 10% of the missing source,” Rhew explained. “That’s a lot of money, and that’s just for copper sulfate. Another copper compound, copper hydroxide, is possibly more widely used. So far, we have only scratched the surface of our understanding of copper’s impact on halocarbon chemistry.”