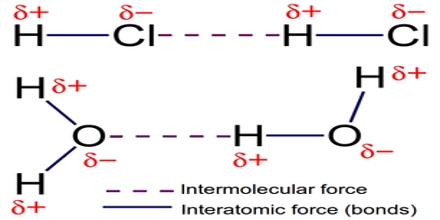
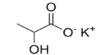
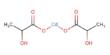
Major objective of this lecture is to discuss on Intermolecular Forces. Covalent bonds exist between atoms within a molecular compound, these covalent bonds are called intramolecular forces. Forces holding entire molecules together are called intermolecular forces. This lecture also focus on Dipole-Dipole Forces and its characteristics. It occurs between polar molecules. Polar molecules have a positive pole and a negative pole so they are called dipoles
Intermolecular Forces