Lymph nodes can play an important role in the success of cancer immunotherapy. Lymph nodes are part of the lymphatic system, which is an important component of the immune system. They contain immune cells, including T cells and B cells, which play a critical role in mounting an immune response to cancer.
Lymph nodes near the tumor are routinely removed during cancer treatment in case they contain metastatic cancer cells. However, new research from UC San Francisco and Gladstone Institutes shows that immunotherapy can activate tumor-fighting T cells in nearby lymph nodes.
The study, published in Cell, suggests that leaving lymph nodes intact until after immunotherapy could improve efficacy against solid tumors, which currently respond to only a small percentage of these newer types of treatments.
Most immunotherapies are aimed only at reinvigorating T cells in the tumor, where they often become exhausted battling the tumor’s cancer cells. But the new research shows that allowing the treatment to activate the immune response of the lymph nodes as well can play an important role in driving positive response to immunotherapy.
Immunotherapy is intended to stimulate the immune response, but when we remove nearby lymph nodes prior to treatment, we’re essentially removing the key locations where T cells live and can be activated.
Matt Spitzer
“This work really changes our thinking about the importance of keeping lymph nodes in the body during treatment,” said Matt Spitzer, Ph.D., an investigator for the Parker Institute for Cancer Immunotherapy and Gladstone-UCSF Institute of Genomic Immunology and senior author of the study.
Lymph nodes are frequently removed because they are the first place metastatic cancer cells appear, and it can be difficult to determine whether the nodes contain metastases without surgery.
“Immunotherapy is intended to stimulate the immune response, but when we remove nearby lymph nodes prior to treatment, we’re essentially removing the key locations where T cells live and can be activated,” Spitzer explained, noting that the evidence supporting lymph node removal comes from older studies that predate the use of today’s immunotherapies.

Aim for the Lymph Nodes, Not the Tumor
Researchers have largely been working under the assumption that cancer immunotherapy works by stimulating the immune cells within the tumor, Spitzer said. But in a 2017 study in mice, Spitzer showed that immunotherapy drugs are actually activating the lymph nodes.
“That study changed our understanding of how these therapies might be working,” said Spitzer. Rather than the immunotherapy pumping up the T cells in the tumor, he said, T cells in the lymph nodes are likely the source for T cells circulating in the blood. Such circulating cells can then go into the tumor and kill off the cancer cells.
Having shown that intact lymph nodes can temper cancer’s hold in mice, Spitzer’s team wanted to know whether the same would prove true in human patients. They chose to design a trial for patients with head and neck cancers because of the high number of lymph nodes in those areas.
The trial included 12 patients whose tumors had not yet spread beyond the lymph nodes. Typically, such patients would have surgery to remove the tumor, followed by additional treatments if necessary. Instead, patients were given a single cycle of atezolizumab (anti-PD-L1), an immunotherapy drug manufactured by Genentech, the trial’s sponsor. Spitzer’s team measured how much the treatment activated the patients’ immune systems a week or two later.
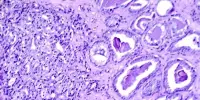
After immunotherapy, each patient’s tumor and nearby lymph nodes were surgically removed and analyzed to see how the immunotherapy affected them. The researchers discovered that after immunotherapy, cancer-killing T cells in the lymph nodes began to activate. They also discovered a higher number of immune cells in the patients’ blood.
Spitzer attributes some of the trial’s success to its design, which allowed the team to collect a large amount of data from a small number of patients by examining tissue before and after surgery and running detailed analyses.
“Being able to collect tissue from surgery shortly after the patients had been given the drug was a really unique opportunity,” he says. “We were able to see, at the cellular level, what the drug was doing to the immune response.”
That kind of insight would be difficult to obtain from a more traditional trial in patients with advanced disease who would not benefit from surgery after immunotherapy.
Metastases Inhibit Immune Response
Another benefit of the study design was that it allowed researchers to compare how the treatment affected lymph nodes with and without metastases, or second cancer growth.
“No one had looked at metastatic lymph nodes in this way before,” said Spitzer. “We could see that the metastases impaired the immune response relative to what we saw in the healthy lymph nodes.”
Spitzer speculates that the therapy may have reduced the activation of T cells in these metastatic nodes. If this is the case, it may help to explain why some immunotherapy treatments perform so poorly. Nonetheless, the therapy stimulated enough T-cell activity in the metastatic lymph nodes to consider leaving them in for a short period of time until treatment ended. “Removing lymph nodes with metastatic cancer cells is probably still important, but doing so before immunotherapy treatment may be throwing the baby out with the bathwater,” Spitzer said.
The current trial’s secondary goal is to see if giving immunotherapy before surgery protects against future tumor recurrence. Researchers won’t know the answer to that until they’ve had a chance to monitor the participants for several years.