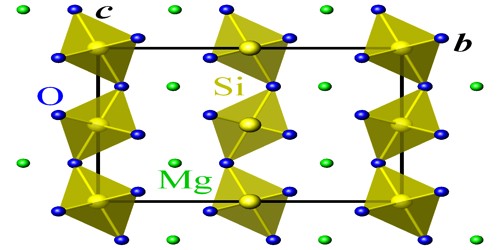
Berlinite (aluminum phosphate, chemical formula AlPO4) is a rare phosphate mineral. It is a rare phosphate mineral first discovered at the Vestana iron mine, Nastum, Sweden. It has the same crystal structure as quartz with a low-temperature polytype isostructural with α–quartz and a high-temperature polytype isostructural with β–quartz. Berlinite can vary from colorless to greyish or pale pink and has translucent crystals.
It was first described in 1868 as an occurrence in the Västanå iron mine, Scania, Sweden and named for Nils Johan Berlin (1812–1891) of Lund University.
General Information
- Category: Phosphate mineral
- Formula: AlPO4
- Crystal system: Trigonal
- Crystal class: Trapezohedral (32).

Properties
It would not be very outstanding except for the fact that it is the only known mineral to be isostructural with quartz. The reason that berlinite is able to have the same structure as quartz is that the aluminum and phosphorus ions are of similar size to silicon ions. Thus the same structure can be achieved since the aluminum and phosphorus can completely replace the silicons without alteration of the quartz structure.
- Color: Colorless, pale gray, maybe pale rose
- Crystal habit: Typically granular to massive
- Fracture: Conchoidal
- Mohs scale hardness: 6.5
- Luster: Vitreous
- Diaphaneity: Transparent to translucent
- Specific gravity: 2.64–2.66
Occurrence
It occurs as a rare mineral in high-temperature hydrothermal or metasomatic deposits. Associated minerals include augelite, attakolite, kyanite, pyrophyllite, scorzalite, lazulite, gatumbaite, burangaite, amblygonite, phosphosiderite, purpurite, apatite, muscovite, quartz, hematite in granite pegmatites. It also occurs with alunite, aragonite, cellophane, crandallite, francoanellite, gypsum, huntite, hydromagnesite, leucophosphite, nesquehonite, niter, and nitrocalcite in the Paddy’s River copper mine in the Brindabella Mountains of Australia.
Information Source: