Schizophrenia often runs in families, implying that it is largely inherited. However, a long-running search for genes responsible for this severe psychiatric condition has yielded only indirect results. Now, gene hunters have discovered for the first time a handful of rare genes that, when mutated, appear to be direct contributors to the disease and may shed light on what goes wrong in the brain of a schizophrenia patient.
The largest genetic study of schizophrenia ever conducted has discovered a large number of specific genes that may play important roles in the psychiatric disorder. To better understand the genes and biological processes underlying schizophrenia, a group of hundreds of researchers from 45 countries examined DNA from 76,755 people with the condition and 243,649 people without it.
The Psychiatric Genomics Consortium study, led by scientists at Cardiff University, found a much larger number of genetic links to schizophrenia than ever before, in 287 different regions of the genome, the human body’s DNA blueprint.
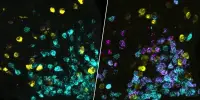
Furthermore, they showed that genetic risk for schizophrenia is seen in genes concentrated in brain cells called neurons, but not in any other tissue or cell type, suggesting it is the biological role of these cells that is crucial in schizophrenia.
We hope the findings in this, and the companion studies, can be used to advance our understanding of the disorder and facilitate the development of radically new treatments. However, those processes are often not straightforward, and a lot of work by other neuroscientists is needed to translate the genetic findings into a detailed understanding of disease mechanisms.
Professor James Walters
The research team say this global study sheds the strongest light yet on the genetic basis of schizophrenia. It is published today in the journal Nature.
“Previous research has shown associations between schizophrenia and many anonymous DNA sequences, but rarely has it been possible to link the findings to specific genes,” said co-lead author Professor Michael O’Donovan, from the Division of Psychological Medicine and Clinical Neurosciences at Cardiff University.
“The present study not only vastly increased the number of those associations, but we have now been able to link many of them to specific genes, a necessary step in what remains a difficult journey towards understanding the causes of this disorder and identifying new treatments.”
Schizophrenia is a serious psychiatric disorder that starts in late adolescence or early adulthood and at any one time affects around one in 300 people worldwide, according to the World Health Organization. In the largest genome-wide association study to date, the research team identified a “substantial increase” in the number of genomic regions associated with schizophrenia. Within these regions, they then used advanced methods to identify 120 genes likely to contribute to the disorder.

As well as being the largest study of its kind, the researchers included more than 7,000 people with either African American or Latino ancestries in what they say is a small step towards making sure advances that come from genetic studies can benefit people beyond those of European ancestries.
Although there are large numbers of genetic variants involved in schizophrenia, the study showed they are concentrated in genes expressed in neurons, pointing to these cells as the most important site of pathology. The findings also suggest abnormal neuron function in schizophrenia affects many brain areas, which could explain its diverse symptoms, which can include hallucinations, delusions, and problems with thinking clearly.
The team’s ability to link to specific genes and areas of biology was enhanced by coordinating their work with a companion study involving many of the same scientists, including those from Cardiff University, led by the Broad Institute of Harvard and MIT, and published in Nature in parallel.
That study looked at mutations that, while very rare, have large effects on the small proportion of people that carry them, and found overlapping genes and overlapping aspects of biology.
Professor James Walters, co-lead author on the Cardiff-led paper and Director of the MRC Centre for Neuropsychiatric Genetics and Genomics at Cardiff University, said: “Whilst people with schizophrenia can recover, many do not respond well to treatments, experience long-term problems with their mental and physical health, as well as impacts on relationships, education, and work.
“We hope the findings in this, and the companion studies, can be used to advance our understanding of the disorder and facilitate the development of radically new treatments. However, those processes are often not straightforward, and a lot of work by other neuroscientists is needed to translate the genetic findings into a detailed understanding of disease mechanisms.”
The Psychiatric Genomics Consortium is funded by the National Institute of Mental Health (NIMH) of the USA and work in Cardiff was additionally supported by the Medical Research Council.
Dr. Joshua Gordon, Director of NIMH, said: “These results, achieved through a global collaboration unprecedented in scope, mark an important step forward in our understanding of the origins of schizophrenia. The findings will allow researchers to focus on specific brain pathways in the ongoing hunt for novel therapies for this serious mental illness.”
This study has demonstrated the importance and power of large samples in genetic studies to gain insights into psychiatric disorders. The team are now seeking to recruit more research participants and build larger, more diverse datasets to further advance our understanding of schizophrenia.
“Landmark study implicates specific genes in schizophrenia”https://t.co/Afcnumqwai
— Marquis_Miami (@Marquis_Miami) May 4, 2022